By Ian Hayman
Charles Linnaeus, the father of modern taxonomy, famously said “A carnivorous plant is against the order of nature as willed by god” when he was presented with specimens and engravings of Dionaea muscipula 1. D. muscipula, the scientific name of the Venus Flytrap, was first exported to Europe in 1768 and became a point of fascination for naturalists at the turn of the 1800s. Flytraps even caught the eye of Charles Darwin, who described them as “the most wonderful plants in the world”1.
Flytraps are just one example of so-called carnivorous plants. Carnivorous plants are found on every continent besides Antarctica and come in forms that are highly varied, both in function and appearance. 60 million years ago there was a duplication event in genome of the common ancestor for all carnivorous plants, resulting in new secondary copies of nutrient uptake genes that are normally expressed in the roots2. This duplication allowed plants that lived in nutrient-poor environments, such as bogs and wetlands, to have the flexibility to express the new gene copies in their leaves. Specialized traps developed from the leaves as a means to collect sparse resources, and selection pressure developed for plants to utilize this new source of nutrients in leaf-based traps. As new species of (now carnivorous) plants developed, further speciation occurred as plants became specialists for certain kinds of prey2. Some of these specialist species, such as flytraps, began to actively catch prey using complex mechanisms similar to our own nervous systems. Other species, such as Nepenthes, developed a passive pitfall system to collect nutrients in the form of insect prey or other detritus.Flytraps are highly evolved to trap prey. In Figure 1, we see a raw image of a trap structure. The cells of the traps are swollen with water, giving the traps a convex shape due to osmotic pressure. There are three needle-like hairs on each side of the trap, that when touched, act as triggers to close the trap (Figure 1A)3. To ensure the trap only closes when prey enters, and not due to random happenstance such as wind or raindrops, at least two signal hairs must be touched. Furthermore, these two hits to the trigger hairs must occur within 30 seconds, otherwise the trap does not close. A calcium-based signal is used to determine when a hair has been hit (Figure 2B)4.
When two signals are received, water within the lobes of the trap is rapidly released, causing the shape to switch from convex to concave (Movie 1)5. The upper ‘teeth’ lining the trap come together in as little as 0.10 seconds to form a cage that keeps insects from escaping. The lobes of the trap close over the course of several seconds. Shifting osmotic pressure is a rapid process, but likely not rapid enough to account for the lightning-fast reflexes of the trap. Flytraps emit a minute magnetic field when they close, which may help to accelerate the osmotic pressure shift by getting all associated plant cells to act rapidly in concert3.
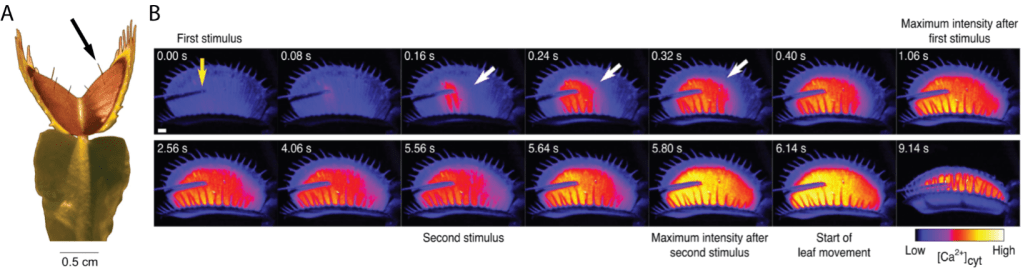
Finally, these specialized plants have one more trick up their proverbial sleeve. Once the trigger hairs are hit and the trap closes, the hairs continue to sense the environment within the trap. If the trap closes around an inorganic object, or the insect prey escapes, the hairs will not be hit additional times. In this case, the plant will simply reopen the trap. If an insect is caught, it will continue to struggle and hit the trigger hairs as a consequence of its movement6. The cells along the rim of the trap will secrete a lipid based ‘glue’, which will make the trap a watertight pouch and completely seal the insect inside7. The trap will then gauge the size of the insect caught through two different mechanisms. First, the more times the trigger hairs are hit, the larger the insect most likely is. The trap will secrete digestive fluids into the sealed pouch, increasing the total fluid and concentration of digestive enzymes in proportion to the number of trigger hair hits6.

Secondly, as the prey dissolves over the course of several days, the insect’s chitin exoskeleton will begin to break down8. Specialized cells within the trap can sense the relative level of digested chitin within the fluid of the trap as a readout for the size of prey captured, as well as its stage of digestion9. The plant can then adjust the amount of digestive fluid secreted into the trap to ensure the entire prey is processed9. After 5-7 days, the trap will reopen. Not all carnivorous plants have such complex prey capture mechanisms2. Genetically distinct plants with pitfall traps are found around the world. Nepenthes are pitfall trap plants with incredible diversity found in southeast Asia (Figure 2). The pitfall trap uses a tube that is designed to trap unwary prey by dropping them into a pit of digestive fluids. These traps have evolved to emit light in the ultraviolet, green, and blue wavebands10. These wavebands are the same used by flowers to attract pollinators and serve to draw insects to traps (Figure 3A)10. This feature is a unique example of convergent evolution, where both carnivorous and non-carnivorous plants developed the same technique to attract insects. In this case, one type of plant uses it to attract pollinators while the other uses it to acquire tasty insect-based snacks.
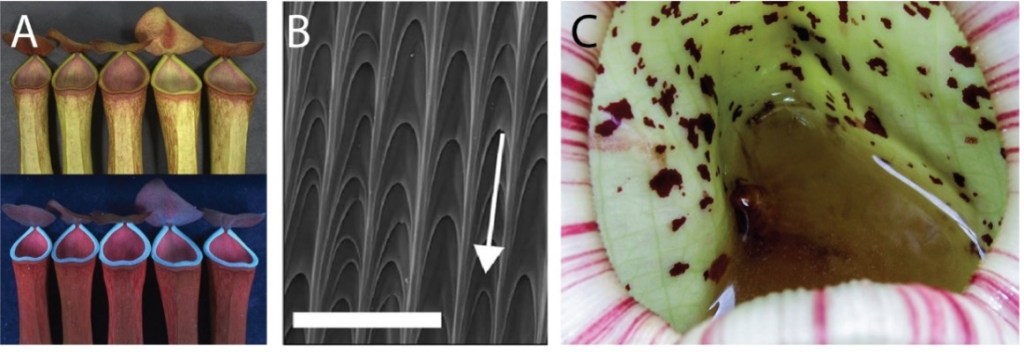
The peristome, or lip of the trap, also has uniquely aligned cells that can wick-up moisture from the air and form a thin skim that repels the oils on insects’ exoskeletons. This makes the rim of the trap very slick, causing insects to slide into the pit when they draw too close (Figure 3B)11. Finally, the digestive fluid in some Nepenthes is highly viscous and sticky, preventing prey from escaping Figure 3C)11. Specialized cells that produce the digestive fluid also produce a number of antifungal compounds to preserve the prey that has been trapped, and the digestive fluid is home to a complex microbiome that assists in digestion12.
Reading about these unique and beautiful plants may inspire you to begin your own collection of them. Unfortunately, carnivorous plants are vanishing from the wild due to climate change and poaching. Luckily, there are numerous groups that use tissue culture techniques and in-house germination to provide a wide variety of these amazing plants without poaching from the natural environment (Pearl River Exotics, California Carnivores, and Sarracenia Northwest to name a few). While all carnivorous plants require specialized care, many can be a relatively easy addition to your house. Flytraps and Sarracenia pitfall traps grow naturally in North America. They can be left outdoors year-round if left in a shallow dish of rain or distilled water. Many Nepenthes species, such as N. maxima and N.ventrata, can be grown on a sunny windowsill if provided with plenty of distilled water and humidity by misting or a pebble tray. Carnivorous plants are a great centerpiece to any plant collection, and now you will be equipped with some fun facts to share with any houseguests! Just don’t name any new plants Audrey II or introduce them to Steve Martin.
References
1 TUCKER, A. The Venus Flytrap’s Lethal Allure. 2021. : < https://www.smithsonianmag.com/science-nature/the-venus-flytraps-lethal-allure-5092361/ >.
2 G, P. et al. Genomes of the Venus Flytrap and Close Relatives Unveil the Roots of Plant Carnivory. Current biology : CB, v. 30, n. 12, 06/22/2020 2020. ISSN 1879-0445. : < https://www.ncbi.nlm.nih.gov/pubmed/32413308 >.
3 FABRICANT, A. et al. Action potentials induce biomagnetic fields in carnivorous Venus flytrap plants. Scientific Reports, v. 11, n. 1, p. 1-7, 2021-01-14 2021. ISSN 2045-23222045-2322. : < https://www.nature.com/articles/s41598-021-81114-w >.
4 SUDA, H. et al. Calcium dynamics during trap closure visualized in transgenic Venus flytrap. Nature Plants, v. 6, n. 10, p. 1219-1224, 2020-10-05 2020. ISSN 2055-02782055-0278. : < https://www.nature.com/articles/s41477-020-00773-1 >.
5 Y, F. et al. How the Venus flytrap snaps. Nature, v. 433, n. 7024, p. 421-425, 01/27/2005 2005. ISSN 1476-4687. : < https://www.ncbi.nlm.nih.gov/pubmed/15674293 >.
6 SCHERZER, S. et al. Insect haptoelectrical stimulation of Venus flytrap triggers exocytosis in gland cells. PNAS, v. 114, n. 18, p. 4822-4827, 2017-05-02 2017. : < https://www.pnas.org/content/114/18/4822 >.
7 AG, V. et al. Venus flytrap biomechanics: forces in the Dionaea muscipula trap. Journal of plant physiology, v. 170, n. 1, p. 25-32, 01/01/2013 2013. ISSN 1618-1328. : < https://www.ncbi.nlm.nih.gov/pubmed/22959673 >.
8 P, P. et al. Secreted major Venus flytrap chitinase enables digestion of Arthropod prey. Biochimica et biophysica acta, v. 1844, n. 2, 2014 Feb 2014. ISSN 0006-3002. : < https://www.ncbi.nlm.nih.gov/pubmed/24275507 >.
9 BEMM, F. et al. Venus flytrap carnivorous lifestyle builds on herbivore defense strategies. Genome Research, 2016-05-04 2016. : < https://genome.cshlp.org/content/early/2016/04/28/gr.202200.115.abstract >.
10 KURUP, R. et al. Fluorescent prey traps in carnivorous plants – Kurup – 2013 – Plant Biology – Wiley Online Library. Plant Biology, v. 15, n. 3, p. 611-615, 2013. : < https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1438-8677.2012.00709.x >.
11 MORAN, J. A.; CLARKE, C. M. The carnivorous syndrome in Nepenthes pitcher plants. Plant Signaling and Behavior, v. 5, n. 6, p. 644-648, 2010. : < http://dx.doi.org/ >.
12 CHAN, X.-Y. et al. Microbiome and Biocatalytic Bacteria in Monkey Cup ( Nepenthes Pitcher) Digestive Fluid. Scientific Reports, v. 6, n. 1, p. 1-10, 2016-01-28 2016. ISSN 2045-23222045-2322. : < https://www.nature.com/articles/srep20016 >.