By Victoria Silvis
Bacteria are an incredibly diverse domain of microorganisms which were among the first organisms living on earth. In order to survive through hundreds of millions of years’ worth of change, these bacteria have learned how to consume and survive on some fascinating and unbelievable substances! Not only have bacteria evolved to eat various components of human diets, but also to eat materials found in our industrialized environment. Here are some fantastic bacteria and the interesting things they feed on.

AmoebaSisters bacterial decomposition cartoon. Available from https://www.amoebasisters.com/parameciumparlorcomics/category/bacteria/4
Halomonas titanicae – The Metal Muncher
The downfall of the Titanic was an iceberg, but the downfall of the shipwreck may be bacteria. Bacteria are ubiquitous, even surviving 2.4 miles below the surface with little light and incredible pressures1. In 2010, a group of scientists from Dalhousie University investigated samples taken from “rusticles”, icicle-shaped rust formations hanging within the ship, for the presence of microorganisms2. One of the isolates found was a novel bacterium dubbed Halomonas titanicae, the first bacterium in the Halomonadaceae family to be found in waters that deep1.The Titanic was constructed of roughly 45,000 tons of steel, iron, and brass3 that now feeds the rapidly replicating bacteria, creating an exponential degradation effect while the bacteria produce ectoine, a stress protectant4,5. Ectoine forms hydrogen bonds with water molecules surrounding the microorganism to aid in modulating the osmotic potential, thus helping H. titanicae survive in high salt environments that would otherwise destroy their cell membranes. Interestingly, the brass in the ship remains untouched, as brass contains copper which serves as an anti-microbial by causing membrane damage and DNA degradation6. While this research is still in its infancy, there are many applications and debates sparking from the discovery of H. titanicae. Understanding how H. titanicae degrades metal can help scientists produce protective coatings on ships or oil rigs and gas pipelines to combat deterioration and future ocean spills7. H. titanicae has the potential to rapidly degrade shipwrecks and other metal-containing structures deep in the ocean, making it favorable to those who want to remove the large amounts of metal waste in our oceans. However, there is an argument8 from historians to kill these microbes in favor of preserving history and adapted marine life.
Ideonella sakaensis – The Plastic Partaker
As a scientist I often look at the vast amount of plastic waste generated in a laboratory and shudder. In 2010, the global plastic waste hit 275 tons, with well over half going straight into landfills9. Polyethylene terephthalate (PET) plastic, patented in 1941, is used to generate most plastic bottles and can take 450 years to decompose in the environment10! There is hope though, as bacteria may come to our rescue. A bacterium called Ideonella sakaiensis was isolated in 2016 at a Japanese waste site and found to digest PET plastic11. However, the bacteria’s degradation rate of the plastic would not be able to mitigate the vast amount of waste produced by humans. In 2018 a group of scientists sought to identify the structure of the evolved PET-degrading enzyme (PETase) but instead accidentally produced a mutant which had improved PET degrading function (visible breakdown in days) and also degraded PEF, a PET substitute12. The improvement did not end here though, as the same team generated a multienzyme system combining the PETase with a MHETase that breaks down MHET, a substance liberated from PET degradation. These two enzymes typically work in tandem to depolymerize PET, however when these two were engineered together, they formed a “super-enzyme”. The scientists found that the combined “super-enzyme” degrades PET six times faster than having the two enzymes work individually13! While there is still work to be done on the front end to minimize plastic waste, I. sakaiensis has the potential to remedy the damage already done.
Pseudomonas putida CBB5 – The Caffeine Connoisseur
Do you ever feel like you’re surviving on caffeine alone? That is exactly what Pseudomonas putida CBB5 can do! In 2011, Pseudomonas putida CBB5 was isolated from flowerbed soil outside a research laboratory at the University of Iowa. P. putida CBB5 was unlike any bacteria seen before as it was the first found to survive solely on caffeine14. Caffeine is comprised of four elements: oxygen, nitrogen, hydrogen, and carbon, and contains three methyl groups. P. putida CBB5 encodes four enzymes which can remove caffeine’s methyl groups via an N-demethylation pathway to eventually form xanthine. Once in the demethylated form, P. putida CBB5 can access internal nitrogen atoms, further breaking xanthine into carbon dioxide and ammonia15. While this newly discovered bacterium is notable for being the first to have the caffeine utilization mechanism parsed out16, P. putida CBB5 also has the potential to revolutionize pharmaceutical and beverage production industries alike.
Pharmaceuticals such as anti-asthmatic or anti-tumor drugs are often generated from derivatives of xanthine. Interestingly, the process of generating xanthine derivatives doesn’t use xanthine as the starting material due to two main challenges: synthesizing xanthine and using expensive reagents17. The ability to use CBB5 enzymes to generate vast quantities of xanthine starting materials from caffeine would revolutionize the production of pharmaceuticals with a xanthine scaffold.
Caffeine-containing beverages, like coffee and tea, often go through a harsh chemical processing to generate decaffeinated versions. The most common way to decaffeinate coffee is to soak the beans in a solvent such as methylene chloride or ethyl acetate, which draws out the caffeine18. Decaffeinated coffee is actually a misnomer, as the beans still contain trace amounts of caffeine, in addition to trace amounts of chemical solvents19. While these methods are FDA-approved19, utilizing caffeine degrading enzymes from P. putida CBB5 may be an easier and potentially safer way to decaffeinate our favorite beverage.
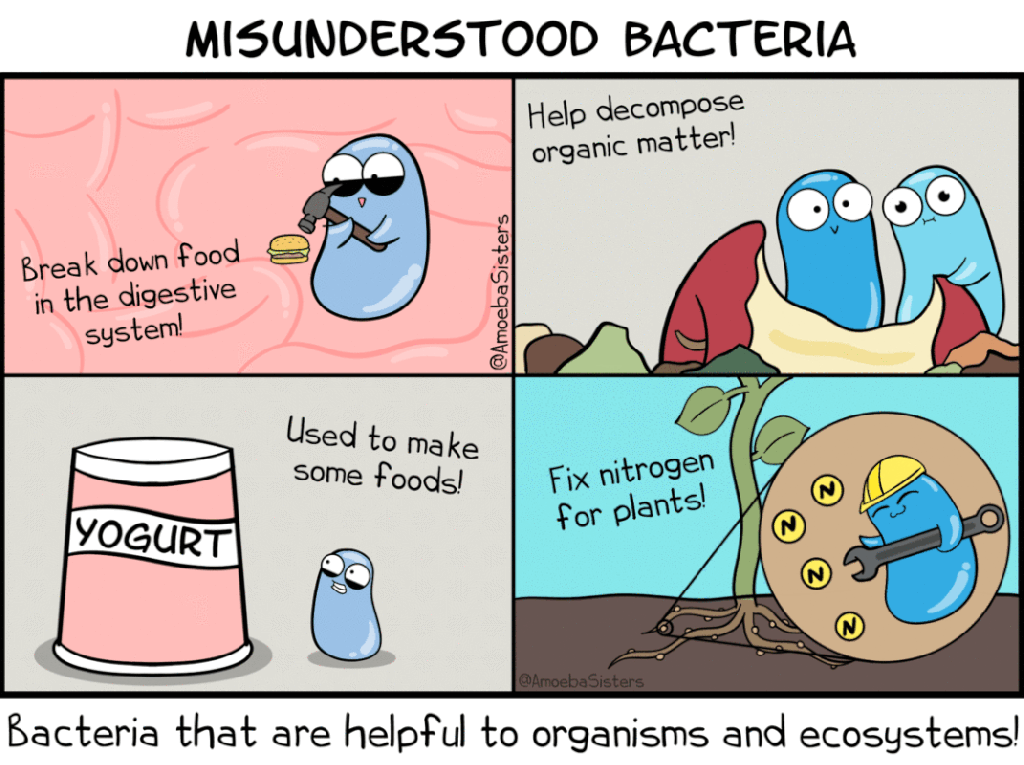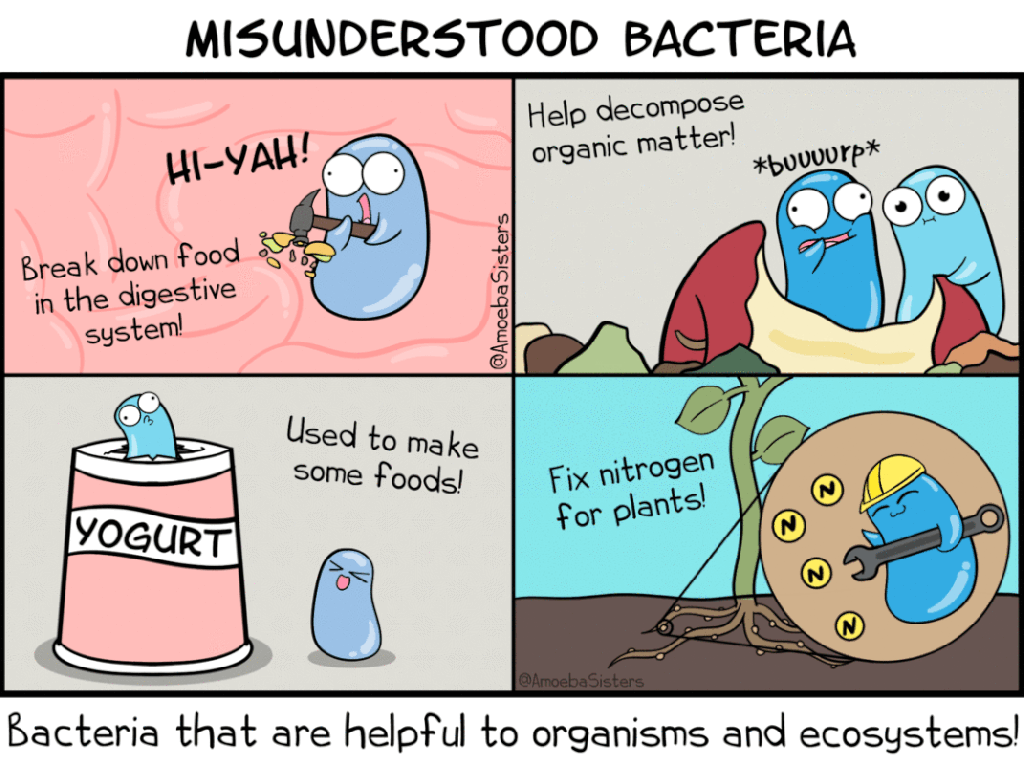
AmeobaSisters misunderstood bacteria GIF. Available from: https://www.amoebasisters.com/gifs.html
I hope you have come to appreciate just how unique microbes in the environment are. In a medically related field, often only host-associated pathogenic or symbiotic bacteria are discussed, but these ubiquitous organisms are everywhere and benefit humans and the environment in some incredible ways!
References
- https://www.nationalgeographic.com/science/article/101210-new-species-bacteria-metal-titanic-wreck-science
- http://www.bbc.com/earth/story/20170310-the-wreck-of-the-titanic-is-being-eaten-and-may-soon-vanish.
- https://ultimatetitanic.com/construction/
- https://www.scienceinschool.org/content/titanic-and-iron-eating-bacteria
- Richter AA, Mais C-N, Czech L, Geyer K, Hoeppner A, Smits SHJ, Erb TJ, Bange G, Bremer E. Biosynthesis of the Stress-Protectant and Chemical Chaperon Ectoine: Biochemistry of the Transaminase EctB. Front Microbiol. 2019 10:2811.
- Mathews S, Hans M, Mücklich F, Solioz M. Contact killing of bacteria on copper is suppressed if bacterial-metal contact is prevented and is induced on iron by copper ions. Appl Environ Microbiol. 2013 Apr;79(8):2605-11.
- https://phys.org/news/2010-12-microscopic-life-aboard-rms-titanic.html
- https://ourworldindata.org/plastic-pollution
- https://www.livescience.com/9079-species-rust-eating-bacteria-destroying-titanic.html
- https://eu.pelacase.com/blogs/news/does-plastic-degrade
- Yoshida S, Hiraga K, Takehana T, Taniguchi I, Yamaji H, Maeda Y, Toyohara K, Miyamotit K, Kamura Y, Oda K. A Bacterium that degrades and assimilates poly(ethylene terephthalate). Science. 2016 March:1196-1199
- Austin HP, Allen MD, Donohoe BS, Rorrer NA, Kearns FL, Silveira RL, Pollard BC, Dominick G, Duman R, El Omari K, Mykhaylyk v, Wagner A, Michener WE, Amore A, Skaf MS, Crowley MF, Thorne AW, Johnson CW, Woodcock HL, McGeehan JE, Beckham GT. Characterization and engineering of a plastic-degrading aromatic polyesterase. PNAS. 2018 May;115(19):E4350-E4357.
- Knott BC, Erickson E, Allen MD, Gado JE, Graham R, Kearns FL, Pardo I, Topuzlu E, Anderson JJ, Austin HP, Dominick G, Johnson CW, Rorrer NA, Szostkiewicz CJ, Copie V, Payne CM, Woodcock HL, Donohoe BS, Beckham GT, McGeehan JE. Characterization and engineering of a two-enzyme system for plastics depolymerization. PNAS. 2020 Oct;117(41):25476-25485.
- https://phys.org/news/2011-05-bacteria-caffeine-food-source.html
- https://phys.org/news/2011-06-caffeine-consuming-bacterium.html
- Summers RM, Mohanty SK, Gopishetty S, Subramanian M. Genetic characterization of caffeine degradation by bacteria and its potential applications. Microb biotechnol. 2015 May;8(3):369-378
- Singh N, Shreshtha AK, Thakur MS, Patra S. Xanthine scaffold: scope and potential in drug development. Heliyon. 2018 Oct;4(10):e00829
- https://www.bbc.com/future/article/20180917-how-do-you-decaffeinate-coffee
- https://www.livescience.com/65278-how-decaf-coffee-is-made.html