By Kaitlin Carson
Are alterations in diet during pregnancy enough to change the course of offspring development? Insights from the Dutch Hunger Winter, a famine that occurred near the end of World War II in which food supplies were severely restricted for a period of 5 months, have helped us address the effects of diet during pregnancy. Though tragic, this famine has provided scientists an opportunity to gleam important epidemiological information about how undernutrition during gestation can alter the development of offspring. By following the lives of children that were born from mothers pregnant during the famine, researchers found that neonates exposed to famine conditions during late gestation were born small for gestational age, and remained small compared to their age-matched counterparts1. Conversely, those exposed to the famine only during early gestation had comparatively normal birth weights but went on to experience higher rates of obesity and cardiovascular disease into adulthood, implying that the programming of adult obesity is more likely to occur during early gestational periods1. The development of the central nervous system (CNS), including crucial brain regions that control feeding behaviors and peripheral organ function, occur during early gestational periods, potentially explaining the observed differences. Beyond obesity, undernutrition during early gestational has also been correlated with impairments in cognition, including attention deficits associated with accelerated aging2. Results from the Dutch Hunger Winter studies contribute to the Developmental Origins of Health and Disease hypothesis, in which exposure to environmental factors, like malnutrition, in utero and in early life can drive the development of disease in adulthood3.
What about maternal overnutrition? Both maternal and general population obesity rates continue to rise rapidly in the United States, with over 40% of Americans considered obese, and near 30% of moms reporting pre-pregnancy obesity. Concurrently, there has been a shift in the composition of the diet we consume, where Americans are eating significantly more calories, particularly from fats. Because of the ever-increasing rates of obesity and changes to diet composition, it is becoming clear that we need to understand the role of maternal overnutrition on fetal programming and long-term health outcomes of these offspring.
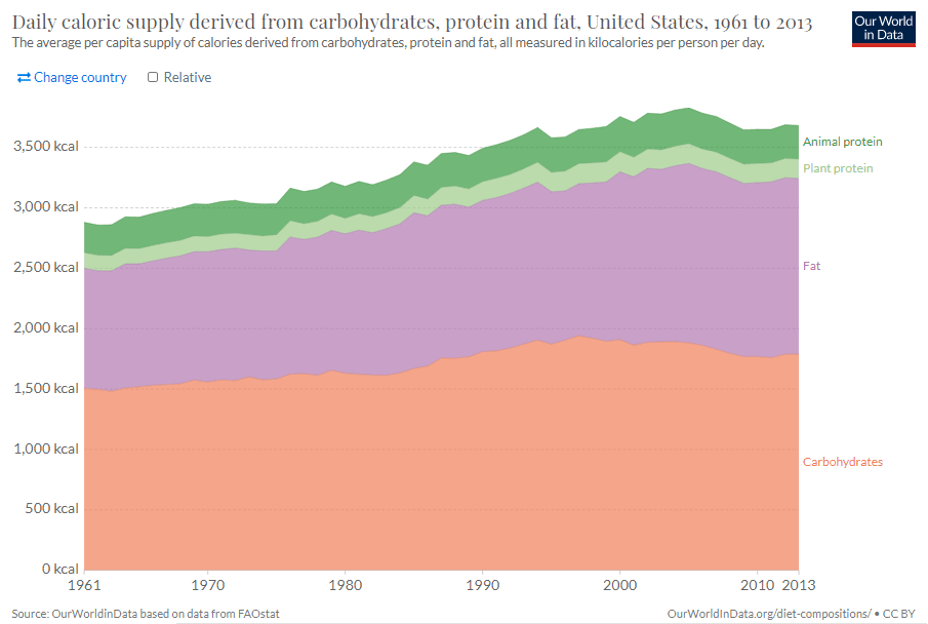
Epidemiological data suggest that maternal obesity is predictive of the development of several health complications in offspring, including childhood obesity, adulthood obesity, type 2 diabetes, and cardiovascular disease4. Exposure to maternal obesity in utero shows a positive correlation with neurodevelopmental and psychiatric complications, such as general deficits in cognition, increased rates of autism, attention deficit hyperactivity disorder (ADHD), mood disorders, anxiety, and eating disorders in offspring 5.
How does diet work to produce these metabolic and neurological changes? Scientists have turned to rodent studies, where pregnant mice and rats are fed a high fat diet (HFD) to model maternal overnutrition. Using the HFD rodent model, researchers have isolated the effects of diet alone on the development of offspring by HFD exposure to only during pregnancy. This is not enough time for the development of obesity in the rats as their gestational period is only 21 days. have found evidence of dysfunction of many organs in the offspring, including the pancreas, liver, and adipose tissue, following maternal HFD exposure, all of which can contribute to the development of obesity and other metabolic complications4. Studies conducted at Penn State College of Medicine have shown that maternal HFD exposure leads to alterations in brain regions that are responsible for controlling function of the gastrointestinal tract6. Dysfunction of these brain regions, including the dorsal motor nucleus of the vagus nerve, been implicated in obesity pathogenesis and functional gastrointestinal disorders7. Other alterations in brain regions have been found following maternal HFD exposure, including elevated immune activation in the hippocampus, a region critical for proper learning and memory5. Lastly, behavioral abnormalities occur after maternal HFD exposure, including reduced sociability and increased anxiety behaviors8. Taken together, results from rodent studies show that maternal HFD exposure is sufficient to induce the development of metabolic and neurologic malfunction in the offspring.
It is becoming clear that maternal diet, whether that is under- or overnutrition, during pregnancy can drive the development of disease in the offspring. Currently, monitoring pregnancy weight gain and food intake are the recommendations given to counteract these potential negative effects. Future work in the maternal nutrition field is needed to better understand if, and how, we can intervene to correct negative developmental outcomes. Is there a critical time frame to intervene to protect development? Are there particular components of the diet that are driving fetal programming? Is there supplementation that can be given during pregnancy or after birth to counteract the effects of malnutrition? Answers to these questions will improve our ability to identify at-risk offspring and take preventative measures.
References
1. Roseboom T, de Rooij S, Painter R. The Dutch famine and its long-term consequences for adult health. Early Hum Dev. Aug 2006;82(8):485-91. doi:10.1016/j.earlhumdev.2006.07.001
2. de Rooij SR, Wouters H, Yonker JE, Painter RC, Roseboom TJ. Prenatal undernutrition and cognitive function in late adulthood. Proc Natl Acad Sci U S A. Sep 28 2010;107(39):16881-6. doi:10.1073/pnas.1009459107
3. Wadhwa PD, Buss C, Entringer S, Swanson JM. Developmental origins of health and disease: brief history of the approach and current focus on epigenetic mechanisms. Semin Reprod Med. Sep 2009;27(5):358-68. doi:10.1055/s-0029-1237424
4. Shrestha N, Ezechukwu HC, Holland OJ, Hryciw DH. Developmental programming of peripheral diseases in offspring exposed to maternal obesity during pregnancy. Am J Physiol Regul Integr Comp Physiol. Nov 1 2020;319(5):R507-r516. doi:10.1152/ajpregu.00214.2020
5. Edlow AG. Maternal obesity and neurodevelopmental and psychiatric disorders in offspring. Prenat Diagn. Jan 2017;37(1):95-110. doi:10.1002/pd.4932
6. Clyburn C, Howe CA, Arnold AC, Lang CH, Travagli RA, Browning KN. Perinatal high-fat diet alters development of GABA(A) receptor subunits in dorsal motor nucleus of vagus. Am J Physiol Gastrointest Liver Physiol. Jul 1 2019;317(1):G40-g50. doi:10.1152/ajpgi.00079.2019
7. Browning KN, Travagli RA. Central nervous system control of gastrointestinal motility and secretion and modulation of gastrointestinal functions. Compr Physiol. Oct 2014;4(4):1339-68. doi:10.1002/cphy.c130055
8. Kang SS, Kurti A, Fair DA, Fryer JD. Dietary intervention rescues maternal obesity induced behavior deficits and neuroinflammation in offspring. J Neuroinflammation. Sep 12 2014;11:156. doi:10.1186/s12974-014-0156-9