By Joseph Cirilo
From the alarm clock in the morning, conversations with co-workers, to the timer in lab informing you that your incubation is finished, sound dominates how we act each and every day. Unfortunately, there is a large population in the US that suffers from hearing loss. According to the National Institute of Deafness and other Communication Disorders (NIDCD), 1 in 8 Americans over the age of 12 experience hearing loss in both ears and 2 to 3 children out of every 1000 have detectable hearing loss in at least 1 ear at birth. However, age remains the greatest predictor of hearing loss. Both men and women age 60-69 are at the greatest risk for hearing loss, though men are two times as likely as women to lose their hearing in their life (1). With the high prevalence of hearing loss, researchers have aimed to understand the mechanisms of how we hear and what causes hearing loss. So, how exactly do we hear?
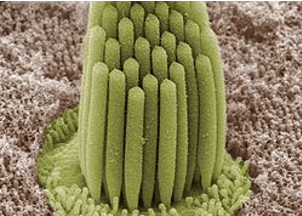
Figure 1: Stereocilia of a Frog inner ear from the Kachar Lab at NIDCD.
The answer to this question is pretty complex. As sound waves enter the ear, they pass through the external auditory canal into the middle ear. Here, the sound waves stimulate the vibration of the tympanic membrane, commonly known as the ear drum. The vibration of this large membrane is then passed through a series of bones, the malleus, incus, and stapes, which serve to “concentrate” the sound waves, increasing their force while minimizing dispersion. These waves are then conducted to the oval window, a membrane that connects the middle ear with the neighboring inner ear. Within the inner ear resides the cochlea, a bony labyrinth filled with a fluid known as endolymph. As the sound waves enter the cochlea, they are transmitted through the endolymph fluid that acts both to amplify the waves and to spread them across the inner ear (2). It is here that the true maestro of hearing, the stereocilia, take over.

Figure 2: Depiction of myosin localization throughout the stereocilia, from Peng et al. 2011.(16)
Stereocilia are hair-like protrusions formed of a tightly bundled parallel actin core, numerous membrane linkers, and important ion channels that help conduct the hearing process. During development, stereocilia form into a staircase-like structure of varying row heights (Figure 1) (3). This structure is imperative to the hearing process throughout an organism’s life, with many studies detailing how altered morphology leads to hearing loss. Charged with maintaining the structure of stereocilia are numerous classes of the molecular motor myosin, including myosin 1C (Myo1C), myosin 3A (Myo3A), myosin 6 (Myo6), myosin 7A (Myo7A), and myosin 15A Myo15A (Figure 2). These myosins generally localize to one of three different areas of the stereocilia: the tip, the tip-link, or the anklet (4-8). The anklet, found at the base of the stereocilia, contains Myo6 which links the membrane of the stereocilia to its actin core(5). Further up the stereocilia, found at the region in which the stereocilia is connected to its smaller neighbor, is the tip-link region. Within this region, Myo7A interacts with a complex of proteins to connect the two protrusions together(9). Mutations in Myo7A have been demonstrated to be associated with dysregulated morphology of stereocilia, resulting in hearing loss (7). Myo1C also can be found localized to this region, though its overall function is still widely unknown (10). Lastly, at the stereocilia tip are Myosins 3A and 15A. Myo15A has been previously shown to help initiate elongation of stereocilia, with mutations leading to unregulated lengths and hearing loss (8). Myo3A also tip localizes, however its overall function is still unknown. Complete loss of Myo3A has been shown to cause extremely long stereocilia, while mutations have been shown to cause both unregulated protrusion size and dynamics(11, 12). Overall, these molecular motors work hand in hand to maintain stereocilia structure and morphology.

Figure 3: Meme describing how MET channels adjust to sound in different environments. Photo Credit: Jeroom, adapted by Joe Cirilo
As the sound wave is conducted through the endolymph, the vibration of the waves causes the stereocilia to move. This movement facilitates the opening of the mechanoelectrical transduction (MET) channels, as the channels are located at the stereocilia tips and are connected to the tip link region of adjacent stereocilia via the same protein linkers as Myo7A (13). As the stereocilia tilt, membrane tension changes and Myo7A is able to walk in a tip-ward direction, opening the channels. As the channels open, K+ ions flood into the hair cells and cause a change in membrane potential. This potential causes an electrical signal to be sent from the auditory nerve to the brain, thus resulting in sound (14). Interestingly, the sensitivity of these channels can also be changed to protect the ear from sounds that are too loud. This process, known as adaptation, can explain why putting the TV on volume 8 is sufficient in the quiet of the night, but way too quiet of a volume when watching in the loud and busy midday (Figure 3). As for pitch, different frequency waves are able to travel different distances within the cochlea. In the case of high pitch sounds such as a flute or piccolo, waves travel a very short distance in the cochlea before being translated into what we hear. However, for low pitch sounds such as a tuba or trombone, those waves travel deeper into the cochlea before being translated (15, 16).
Overall, how we hear is still a complicated process with many unanswered questions. However, researchers are actively investigating not only how we hear, but also how hearing loss occurs in hopes of finding therapeutics to restore hearing. Current research in Myo15A by the Friedman and Belyantseva groups at the NIDCDC, Frolenkov group at the University of Kentucky, and Bird group at the University of Florida, have aimed to understand how Myo15A facilitates stereocilia production and maintenance, while studies by the Yengo Lab here at Penn State College of Medicine are actively investigating the role of Myo3A in stereocilia, as well as the effects of many deafness mutations. Lastly, the Boye group at University of Florida has shown promising results of Myo7A gene therapy as treatment for hearing loss. With these researchers actively working hard, there is no doubt that many of these questions will get answered. Doesn’t that “sound” great?
- Disorders NIoDaOC. Quick Statistics About Hearing [Web]. NIH: NIH; 2016 [updated December 15, 2016; cited 2020 July 7]. Available from: https://www.nidcd.nih.gov/health/statistics/quick-statistics-hearing.
- Heine PA. Anatomy of the ear. Vet Clin North Am Small Anim Pract. 2004;34(2):379-95. Epub 2004/04/06. doi: 10.1016/j.cvsm.2003.10.003. PubMed PMID: 15062614.
- Manor U, Kachar B. Dynamic length regulation of sensory stereocilia2008;19(6):502-10. doi: 10.1016/j.semcdb.2008.07.006.
- Lin T, Greenberg MJ, Moore JR, Ostap EM. A Hearing Loss-Associated myo1c Mutation (R156W) Decreases the Myosin Duty Ratio and Force Sensitivity2011;50(11):1831-8. doi: 10.1021/bi1016777.
- Wang J, Shen J, Guo L, Cheng C, Chai R, Shu Y, Li H. A humanized mouse model, demonstrating progressive hearing loss caused by MYO6 p.C442Y, is inherited in a semi-dominant pattern. Hearing Research. 2019;379:79-88. doi: 10.1016/j.heares.2019.04.014.
- Dosé AC, Hillman DW, Wong C, Sohlberg L, Lin-Jones J, Burnside B. Myo3A, One of Two Class III Myosin Genes Expressed in Vertebrate Retina, Is Localized to the Calycal Processes of Rod and Cone Photoreceptors and Is Expressed in the Sacculus. Molecular Biology of the Cell. 2003;14(3):1058-73. doi: 10.1091/mbc.e02-06-0317.
- Calabro KR, Boye SL, Choudhury S, Fajardo D, Peterson JJ, Li W, Crosson SM, Kim M-J, Ding D, Salvi R, Someya S, Boye SE. A Novel Mouse Model of MYO7A USH1B Reveals Auditory and Visual System Haploinsufficiencies. Frontiers in Neuroscience. 2019;13. doi: 10.3389/fnins.2019.01255.
- Rehman AU, Bird JE, Faridi R, Shahzad M, Shah S, Lee K, Khan SN, Imtiaz A, Ahmed ZM, Riazuddin S, Santos-Cortez RLP, Ahmad W, Leal SM, Riazuddin S, Friedman TB. Mutational Spectrum ofMYO15Aand the Molecular Mechanisms of DFNB3 Human Deafness. Human Mutation. 2016;37(10):991-1003. doi: 10.1002/humu.23042.
- Yu IM, Planelles-Herrero VJ, Sourigues Y, Moussaoui D, Sirkia H, Kikuti C, Stroebel D, Titus MA, Houdusse A. Myosin 7 and its adaptors link cadherins to actin. Nature Communications. 2017;8(1):15864. doi: 10.1038/ncomms15864.
- Mecklenburg KL, Freed SA, Raval M, Quintero OA, Yengo CM, O’Tousa JE. Invertebrate and vertebrate class III myosins interact with MORN repeat-containing adaptor proteins. PLoS One. 2015;10(3):e0122502. PubMed PMID: 25822849.
- Dantas VGL, Raval MH, Ballesteros A, Cui R, Gunther LK, Yamamoto GL, Alves LU, Bueno AS, Lezirovitz K, Pirana S, Mendes BCA, Yengo CM, Kachar B, Mingroni-Netto RC. Characterization of a novel MYO3A missense mutation associated with a dominant form of late onset hearing loss. Scientific Reports. 2018;8(1). doi: 10.1038/s41598-018-26818-2.
- Lelli A, Michel V, Boutet De Monvel J, Cortese M, Bosch-Grau M, Aghaie A, Perfettini I, Dupont T, Avan P, El-Amraoui A, Petit C. Class III myosins shape the auditory hair bundles by limiting microvilli and stereocilia growth2016;212(2):231-44. doi: 10.1083/jcb.201509017.
- Grati M, Kachar B. Myosin VIIa and sans localization at stereocilia upper tip-link density implicates these Usher syndrome proteins in mechanotransduction2011;108(28):11476-81. doi: 10.1073/pnas.1104161108.
- Yue X, Sheng Y, Kang L, Xiao R. Distinct functions of TMC channels: a comparative overview. Cell Mol Life Sci. 2019;76(21):4221-32. Epub 2019/10/05. doi: 10.1007/s00018-019-03214-1. PubMed PMID: 31584127.
- Lorimer T, Gomez F, Stoop R. Mammalian cochlea as a physics guided evolution-optimized hearing sensor. Sci Rep. 2015;5:12492. Epub 2015/07/29. doi: 10.1038/srep12492. PubMed PMID: 26216397; PMCID: PMC4516981.
- Peng AW, Salles FT, Pan B, Ricci AJ. Integrating the biophysical and molecular mechanisms of auditory hair cell mechanotransduction. Nature Communications. 2011;2(1):523. doi: 10.1038/ncomms1533.