By Rahul Nachnani
Millions of patients are struggling with treatment-resistant psychiatric diseases including depression, post-traumatic stress disorder (PTSD), and addiction. These folks often go through numerous rounds of medications, therapies, providers, and are understandably frustrated with the lack of progress in their mental health and recovery. In an ideal setting, rather than months of ring-around-the-pharmacy, patients could walk into a provider’s office and take a treatment that by the end of that day creates a significant reduction in their symptoms and stress for months. These appealing outcomes are why psychedelics are being studied as medicine. Psychedelic drugs are psychoactive, usually derived from plants, and function to “alter perception, cognition, or mood”.1 Our limited understanding of these high-potency chemicals has been stifled throughout history. In the past thirty years, research on psychedelics as medicine has blossomed as a form of hope for patients in need. To better understand how we can use psychedelics today, it’s important to first discuss the use of psychedelics in the past (the first introduction). Then, let’s take a trip through the laboratory to figure out how psychedelics are now used in clinical trials to address long-standing pharmacotherapeutic needs (the “re-introduction”).
Psychoactive plants and fungi have served as a core arm of indigenous spirituality longer than written history can estimate. Indigenous communities across North and South America have used psychedelics in religious ceremonies, medical procedures, and when seeking spiritual insight.2-4 Much of the history and practices of Latin American tribes were lost to Christianization, and spiritual use of sacred plants was condemned for hundreds of years following.3 Some traditions remained and were shared with curious foreign scientists. The Western world medicalized psychedelics around the 1940s when Albert Hofman published his group’s synthesis of Lysergic Acid Diethylamide (LSD), a chemical derivative of ergot fungus which grows on wheat.2 This derivation instilled a renaissance-like movement and led artists, authors, scientists, doctors, and the public to become curious about the new experiences that psychedelics granted. However, the renaissance quickly came to an end due to a variety of political, militaristic, and xenophobic powers.4 Twenty years after Hofmann’s discovery, the United Nations Convention of 1971 outlawed psychedelic drugs, criticizing their lack of purpose and potential to harm. Despite the criticism, medical research did not forsake these sacred plants as researchers across the earth have conducted mechanism-driven studies and clinical trials to discover modern uses for this ancient medicine. Psychedelics, though, are not a homogenous group, so before understanding their potential in medicine, it’s good to get a handle on a few main origins.

Table 1: Cheat sheet of the origins and chemical derivatives of five different commonly used plant and fungal psychedelics from around the world. Photo Credits: Psilocybin https://hub.jhu.edu/2016/12/01/hallucinogen-treats-cancer-depression-anxiety/ Peyote: https://www.123rf.com/photo_71983641_pink-blossom-of-lophophora-williamsii-peyote-cactus.html Ololiuqui: https://www.britannica.com/plant/ololiuqui Caapi vine: https://eternityinabox.com/shop/white-banisteriopsis-caapi-ayahuasca-whole-vine/
While there are a variety of classes of psychedelics, the drugs most studied for therapy are all “serotonergic agonists.” This means that the drugs activate the same receptors as serotonin, a naturally produced neurochemical by our bodies, and induce effects of serotonin, altering a variety of traits like mood, stress, memory, and even blood pressure. Most psychedelic drugs stimulate one subtype of serotonin receptors: the serotonin 2A receptor (5-HT2A) in the brain and body.4 These 5-HT2A receptors are activated when one ingests a hallucinogen and cause chemical and structural changes in the brain, mediating the short-term and long-term effects of psychedelics. While the euphoric and mind-bending short-term effects are the culprits of the popularity of psychedelics amongst recreational users, the long-term structural changes are sought after by clinicians and researchers as a potential method to create lasting changes in brain chemistry. In a 2017 Nature Neuropsychopharmacology article by authors Carhart-Harris and Goodwin, the researchers compare psychedelic effects at 5-HT2A receptors to Selective Serotonin Reuptake Inhibitor (SSRI) activity at serotonin 1A (5-HT1A) receptors.6 SSRIs like Prozac (Fluoxetine) and Zoloft (Sertraline) are the prototypical antidepressants used for many different psychiatric illnesses. It is standard practice to compare a potential treatment to the current “gold-standard” treatment (in this case, SSRIs), and clinical trials use this comparison to understand both the beneficial and harmful effects of psychedelics. Pharmacology is not the only way researchers are trying to understand psychedelics, though, and recent innovation in brain imaging helps us know more about how psychedelics affect our perception.
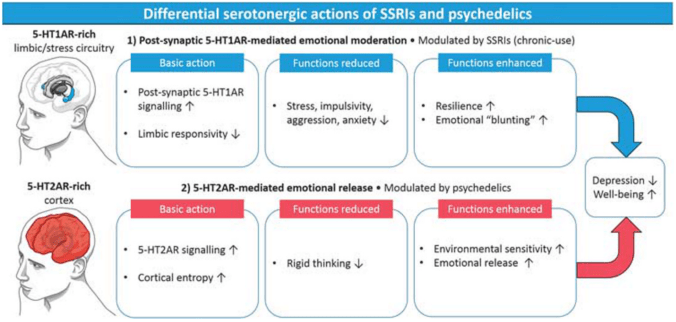
Figure 1: Carhart-Harris and Goodwin compare and contrast the methods by which SSRIs and psychedelics could induce anti-depressant effects and promote well-being in patients. A major difference is that the anti-depressant effects of psychedelics can be seen with as little as a single use, while SSRIs take months of daily use to exert their full effects. Because psychedelics have a different mechanism than SSRIs, more work must be done to better understand their dangers and benefits as therapy. Photo Credit: Carhart-Harris and Goodwin, Neuropsychopharmacology 2017.
If you looked away from reading this article for a minute and focused on, well, nothing, your brain would show activity in the default mode network (DMN), also called the “resting network”. Various alterations in the DMN are implicated in neurological diseases like Alzheimer’s disease, autism, schizophrenia, PTSD, generalized anxiety, and addiction. In a mental illness like PTSD, past trauma and adverse experiences may alter the DMN to more easily re-route neutral stimuli to the areas of our brains which react to stress. Psychedelic usage alters the DMN and reduces the connectivity between the DMN and other neural circuits we use to interpret the world around us. 7 However, these alterations to neural connectivity are powerful and sometimes difficult to predict. For patients struggling with PTSD, one session with a psychedelic along with the help of an experienced mental healthcare provider can rewire the patient’s brain to reduce the uncontrollable stress that comes with reliving a traumatic experience7. This is an exciting potential mechanism for the effects of psychedelics; however, it is important to emphasize that the most benefit of any psychedelic treatment will come from the combination of a safe environment and a trusted, experienced mental healthcare provider. These providers can guide a patient through novel and intense experiences along with facilitate ways to interpret experiences and put new ideas into practice. A guide-less psychedelic experience runs the risk of prolonged and intense negative side effects (ever heard of a “bad trip”?), which is the main reason why this high-power class of drugs must be administered responsibly, and why more research is needed.
There is unrealized opportunity for psychedelics in psychiatric illness. Many gaps which have yet to be filled by psychiatric therapy might be addressed in the future with psychedelic medicine. More importantly, as we better understand the derivatives of an ancient medicine, we must take it upon ourselves to pay attention and listen to the traditions of cultures at risk of becoming erased. There would be no potential psychedelic medicine without indigenous healers sharing their expertise. Psychedelic research is a prime example of applying past medicine to current illnesses and may serve as a model for drug design and development in the future.
Sources:
- https://www.nature.com/articles/1380423
- Evans Schultes, Hofmann and Ratsch (1998) Plants of the Gods. Healing Arts Press
- https://theconversation.com/discovering-hallucinogenic-mushrooms-in-mexico-115033
- https://www.rcpsych.ac.uk/docs/default-source/members/sigs/spirituality-spsig/ben-sessa-from-sacred-plants-to-psychotherapy.pdf?sfvrsn=d1bd0269_2
- https://maps.org/research-archive/w3pb/2002/2002_riba_6009_1.pdf
- https://www.nature.com/articles/npp201784
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6787540/