By Stephanie Baringer
It is always exciting for scientists to see their research interests published in high impact journals, such as Nature, Cell, and Science. One can imagine my excitement when two articles that focused on the blood brain barrier (BBB) and Alzheimer’s disease (AD) were published in Nature within about a month of each other. Despite the different studies coming from separate labs on opposite sides of the country, the results from each support the findings from the other. These studies highlight the importance of understanding how science in the clinic and at the bench can be interwoven to aid in further discovery and therapeutic progress.
As a brief background, the BBB is the barrier that separates blood vessels and brain tissue.1 This barrier is extremely important in regulating the transport of a variety of molecules, ions, and nutrients and in preventing the entry of toxic compounds to the brain. The BBB is primarily made up of three types of cells: endothelial cells, pericytes, and astrocytes. Endothelial cells (ECs) are the first line of defense and encircle the blood vessels. They are responsible for the transport of molecules and ions. Pericytes sit on the ECs and aid in EC tight junction and gap junction strength. Pericytes can also exert control of capillary diameter by contracting.2 At the BBB, astrocytes are glial cells that provide a link between neurons and the blood vessels. Numerous disorders have been linked to BBB dysfunction, notably AD and other neurodegenerative diseases.3
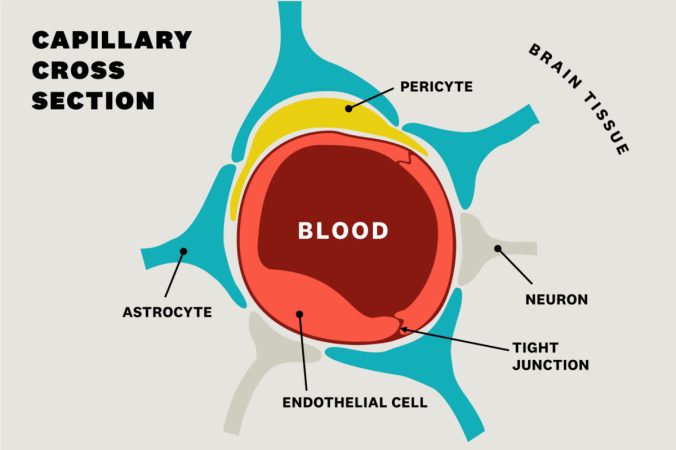
Photo credit: Leigh Hopper, USC News
The connection between vascular dysfunction and dementia is increasingly gaining recognition. While the increased risk associated with the APOE4 variant of the APOE gene have been extensively studied in AD, the mechanistic relationship with the BBB is still being unveiled. Along this line of investigation, the BBB breakdown may be an early biomarker for AD and APOE4 leads to overall BBB and specifically pericyte breakdown.3,4 However, the cognitive effects of such BBB breakdown were unknown until recently. The Zlokovic group used a case-control clinical study design to understand how breakdown of the BBB can contribute to and predict APOE4-associated cognitive decline in AD, regardless of disease pathology.5 The researchers were blinded to genotype and able to distinguish APOE4 carriers based on regional BBB breakdown in the hippocampus and medial temporal lobe that was independent of the hallmarks of AD pathology, amyloid-β (Aβ) or tau pathology. While BBB breakdown was present in cognitively unimpaired APOE4 carriers, it was more severe in those who had cognitive impairment. In support of these observations, the researchers found increased levels of soluble platelet-derived growth factor receptor-β (PDGFRβ), a pericyte injury marker, and other markers of BBB breakdown in CSF. These biomarkers were predictive of cognitive decline in APOE4 carriers only. Lastly, the activation of the CypA-MMP9 pathways by pericytes leads to the breakdown of the BBB, and thus, the CSF levels of cyclophilin A (CypA) and matrix metalloproteinase-9 (MMP9) were increased in APOE4 carriers with cognitive impairment. Montagne et al. concluded that the breakdown of the BBB predicts APOE4-associated cognitive decline, independent of disease pathology.5
Amyloid deposits along the vasculature lead to cerebral amyloid angiopathy (CAA), which leads to impaired BBB function. Over 90% of AD patients also develop CAA, and APOE4 is the strongest genetic risk factor for both. Related to Zlokovic’s clinical methods, the Tsai group primarily relied on data collected from a cellular model of the BBB.6 The researchers created a cell culture model, termed iBBB, using the three cell types that make up the BBB, all derived from induced pluripotent stem cells (iPSCs). Consistent with in vivo findings, their iBBB model accumulated minimal exogenous Aβ compared to the AD mutated neurons. However, iBBB with APOE4 mutations accumulated more Aβ. After performing a screen of various combinations of APOE4 and APOE3 (APOE3 is the normal variant of APOE) ECs, pericytes, and astrocytes, only cultures with APOE4 pericytes displayed the increase in Aβ accumulation. To dig further into the potential mechanism, the researchers found that APOE and one of its transcription factors, NFAT, were upregulated in the APOE4 pericytes. Blanchard et al. then inhibited calcineurin, the enzyme that activates NFAT, and found that doing so reduced the expression of APOE and reduced the amount of Aβ deposition.6 Overall, Blanchard et al. concluded that BBB pericytes play a crucial role in APOE-associated CAA, and thus BBB breakdown, through the calcineurin-NFAT signaling pathway.
The publication of these two studies in Nature in such a short amount of time emphasizes the importance of an up-and-coming perspective of AD progression in relation to BBB breakdown. In their discussions, both papers hint at the potential of therapeutics to target the accumulation of Aβ and breakdown of the BBB that precedes cognitive decline. Montagne et al. propose exploration of CypA inhibition to prevent pericyte breakdown, and Blanchard et al. suggest calcineurin inhibition to prevent APOE overexpression and Aβ accumulation. While these potential therapeutic targets have not been further explored as of yet, these parallel studies shed light on the BBB breakdown mechanism of the disease progression. As scientists, it is important to remember how strong the connection between bench and clinical data is. After all, many of us conduct research in hopes to improve patient therapies and care.
References:
(1) Daneman, R.; Prat, A. The Blood–Brain Barrier. Cold Spring Harb Perspect Biol 2015, 7 (1). https://doi.org/10.1101/cshperspect.a020412.
(2) Hall, C. N.; Reynell, C.; Gesslein, B.; Hamilton, N. B.; Mishra, A.; Sutherland, B. A.; O’Farrell, F. M.; Buchan, A. M.; Lauritzen, M.; Attwell, D. Capillary Pericytes Regulate Cerebral Blood Flow in Health and Disease. Nature 2014, 508 (7494), 55–60. https://doi.org/10.1038/nature13165.
(3) Sweeney, M. D.; Sagare, A. P.; Zlokovic, B. V. Blood–Brain Barrier Breakdown in Alzheimer Disease and Other Neurodegenerative Disorders. Nat Rev Neurol 2018, 14 (3), 133–150. https://doi.org/10.1038/nrneurol.2017.188.
(4) Halliday, M. R.; Rege, S. V.; Ma, Q.; Zhao, Z.; Miller, C. A.; Winkler, E. A.; Zlokovic, B. V. Accelerated Pericyte Degeneration and Blood–Brain Barrier Breakdown in Apolipoprotein E4 Carriers with Alzheimer’s Disease. J Cereb Blood Flow Metab 2016, 36 (1), 216–227. https://doi.org/10.1038/jcbfm.2015.44.
(5) Montagne, A.; Nation, D. A.; Sagare, A. P.; Barisano, G.; Sweeney, M. D.; Chakhoyan, A.; Pachicano, M.; Joe, E.; Nelson, A. R.; D’Orazio, L. M.; Buennagel, D. P.; Harrington, M. G.; Benzinger, T. L. S.; Fagan, A. M.; Ringman, J. M.; Schneider, L. S.; Morris, J. C.; Reiman, E. M.; Caselli, R. J.; Chui, H. C.; Tcw, J.; Chen, Y.; Pa, J.; Conti, P. S.; Law, M.; Toga, A. W.; Zlokovic, B. V. APOE4 Leads to Blood–Brain Barrier Dysfunction Predicting Cognitive Decline. Nature 2020, 1–6. https://doi.org/10.1038/s41586-020-2247-3.
(6) Blanchard, J. W.; Bula, M.; Davila-Velderrain, J.; Akay, L. A.; Zhu, L.; Frank, A.; Victor, M. B.; Bonner, J. M.; Mathys, H.; Lin, Y.-T.; Ko, T.; Bennett, D. A.; Cam, H. P.; Kellis, M.; Tsai, L.-H. Reconstruction of the Human Blood–Brain Barrier in Vitro Reveals a Pathogenic Mechanism of APOE4 in Pericytes. Nature Medicine 2020, 26 (6), 952–963. https://doi.org/10.1038/s41591-020-0886-4.