By Rebecca Fleeman, PhD
The following is a synopsis of my PhD thesis that I defended on January 19, 2023, titled Alzheimer’s Disease Risk Conveyed by Apolipoprotein E ε4: Immunometabolism and the Brain-Body Connection. Thank you to LTS for the opportunity to share my PhD findings in a unique way to reach more people!
The Problem:
Alzheimer’s disease (AD) is a neurodegenerative disease resulting in memory loss and cognitive impairment that affects over 50 million people globally. Nearly everyone knows someone who has been affected by AD or dementia, since 1 in 9 seniors over the age of 65 develop AD1. When someone develops dementia symptoms from AD they begin to lose the ability to complete familiar tasks, plan and problem solve, and keep track of time. These cognitive detriments begin subtly, but accrue over time, becoming devastating to friends and family who can no longer be remembered by those with AD. To add to this hardship, there are currently no curative treatments for AD and the molecular mechanisms of disease pathogenesis remain incompletely understood (Figure 1).
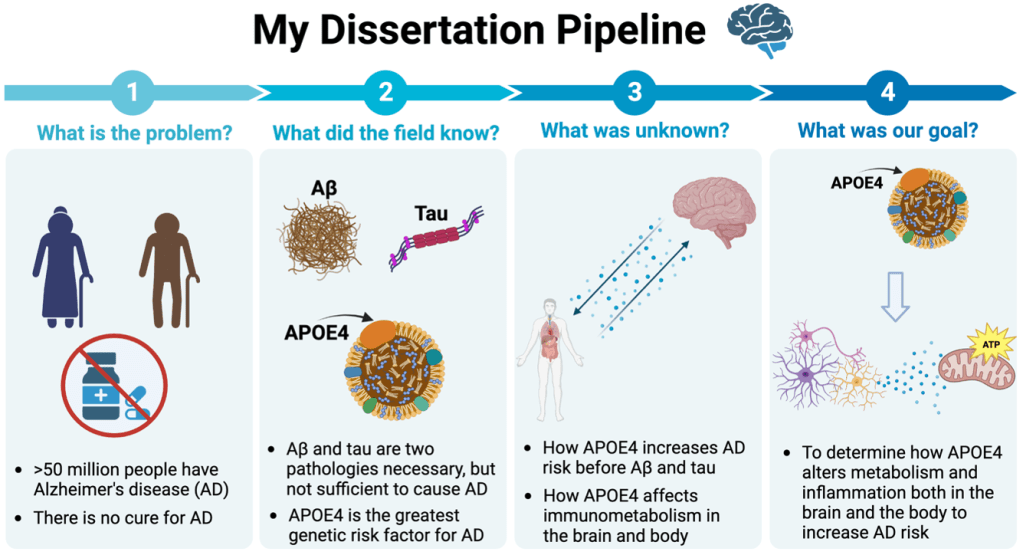
What Was Known Before My Findings:
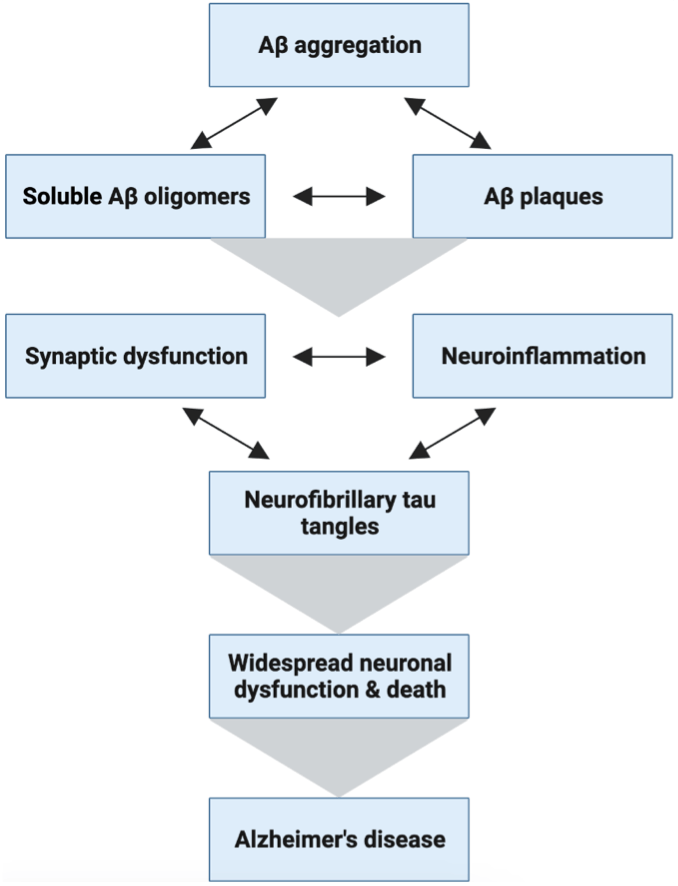
While not fully understood, the pathogenesis of AD has been highly studied and involves the development of the hallmark protein pathologies (proteinopathies) amyloid β (Aβ) plaques and neurofibrillary tau tangles. These two proteinopathies, while once only detectable postmortem, can now be reliably detected using brain imaging techniques (and blood-based biomarkers for these pathologies are close to approval, see my previous LTS article!). Since Aβ deposition precedes both tau pathology and cognitive decline seen in AD, a large portion of the field has focused on this Amyloid Cascade Hypothesis (Figure 2). Both of the recently approved AD therapeutics (non-curative) are based on this hypothesis and target Aβ2–6. Besides these key pathologies, several genetic risk factors have also been evaluated for their impact on AD.
The ε4 variant of apolipoprotein E (APOE) is the strongest and most common genetic risk factor for AD. APOE is a lipid transport protein, important in the brain for shuttling cholesterol between cells that can be incorporated for injury repair, membrane maintenance, and to aid neuronal signaling. There are 3 variants that humans possess, APOE2, APOE3, and APOE4, with APOE3 being the most prevalent, risk-neutral variant, APOE2 being the least common, risk averse variant, and APOE4 being carried by 10-20% of the population, increasing risk for AD by 4-14 fold, and decreasing the age of disease onset.7–10 While the mechanism of APOE4’s conveyed risk for AD is incompletely understood, APOE4 is known to promote inflammation, dysregulate metabolism, and increase protein misfolding and aggregation, each outcome of which contributes to accelerating AD pathology.11–19
APOE4 was first discovered in the 1970s, known for its effects on dysregulating lipid metabolism and creating risk for cardiovascular disease8. It wasn’t until the 1990s that APOE4 became known for its ability to increase risk for AD7,9,20. Previous research has mainly occurred in silos, where some laboratories study the peripheral body effects of APOE4, focusing on its risk for cardiometabolic diseases, or separately on how APOE4 interacts with hallmark pathologies of AD, Aβ plaques and neurofibrillary tau tangles, in the brain. Thus, a key gap remaining in the field is connecting the peripheral and central immune and metabolic effects of APOE4, in the context of healthy and diseased aging, to better identify how APOE4 creates an AD-prone environment.
My Main Question:
How does APOE4 alter metabolism and inflammation both in the brain and the body to increase AD risk?
What I Did to Answer the Question:
During my PhD, I used murine in vivo, ex vivo, and in vitro methods (live mouse experiments, postmortem mouse experiments, and cell culture experiments), followed by multivariate modeling to better understand relationships between immune signaling and metabolic function (Figure 3A-C). I measured several peripheral metabolic factors in mice carrying either humanized APOE3 or APOE4 alleles, then quantified the immune signaling in their brain (Figure 3A). Using multivariate modeling, I compared the effects of APOE genotype, sex, and age on these measurements, as well as connected the changes in immune signaling to changes in metabolism (Figure 3C).
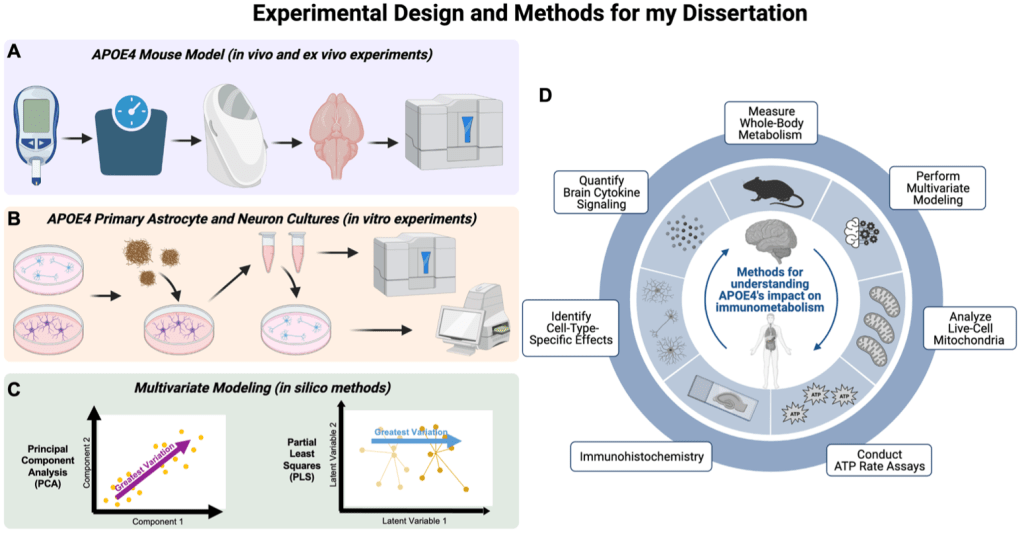
I additionally utilized primary murine cell culture models to better understand the mechanism of changes in immunometabolism due to APOE genotype. I created cultures of neurons and astrocytes, two main brain cell types, from APOE3 and APOE4 mice, and quantified how metabolism and immune signaling of these cells changed when treated with AD proteinopathy (Figure 3B-C). Because astrocytes are key immune and metabolic support cells to neurons, I treated the astrocytes with Aβ for three days, then I drew up the media from the astrocytes, which now contained the reactionary secretions from the Aβ treatment. I then used half that media to run experiments on, and the other half was used to treat neurons, so that I could observe how the astrocytic reactions to Aβ affect neuronal health.
Key Take Aways:
AD, APOE4, and Aβ pathology have been well studied and characterized over several decades, and yet, no disease-modifying therapy for AD has come to market. Thus, looking beyond the Amyloid Cascade Hypothesis is essential to better understanding how APOE4 increases risk and decreases the age of onset of AD, so that we can identify successful therapeutic targets for AD6,21.
Overall, my dissertation leverages murine in vivo, ex vivo, and in vitro methods to showcase how the greatest genetic risk factor for AD, APOE4,
(1) creates an altered environment in the brain and body with aging, prior to onset of AD proteinopathies22;
(2) alters astrocytic glucose metabolism, in turn altering cytokine secretion, ATP production, and downstream neuronal health23; and
(3) increases cholesterol in astrocytes, with the potential to alter tau internalization and the inflammatory and metabolic responses that accompany AD proteinopathies24 (Figure 4).
While I cannot deep dive into every finding I made, I do want to highlight some really cool things I was able to do. The first being, using the concentrations of cytokines (little signaling molecules in the brain) in the brains of mice, I was able to use multivariate modeling to predict peripheral metabolic outcomes. I legitimately could tell you how fat a mouse was, just based off of brain signaling. Not only this, I was also able to predict mouse genotype, sex, and age from cytokines. The conscious reader will likely say “Cool!!….so what? Why should I care?”. Well, finding relationships between the brain and body is essential to better understanding disease pathogenesis, risk factors, and biomarkers, and these are all things you should care about! Recall that 1 in 9 seniors develops AD. If you could have a better understanding of how to modulate your lifestyle choices to decrease AD risk, wouldn’t you want to know?

Taken together, these findings highlight the detrimental effects of APOE4, with and without AD proteinopathies present. Understanding dysregulation of immunometabolism is a newer avenue in the field of AD research, and these data add to the growing body of APOE4 immunometabolism literature, showcasing the necessity of better characterizing immune signaling and metabolic dysfunction in multiple cell types individually, as well as in whole regions of the brain, to fully understand why APOE4 accelerates and amplifies AD progression.
The Implications of My Work:
At the time this article was published, only 6 approved therapies exist for Alzheimer’s patients. Four of those only treat the dementia symptoms by improving neuronal signaling. The other two, both approved within the past 2 years, are immunotherapies targeting Aβ. While these two therapies successfully reduce the amount of Aβ in the brain, the observed reduction does not stop or reverse the cognitive decline and memory loss seen in AD patients.
The immune signaling and metabolic profiles I identified in the hippocampus of APOE3 and APOE4 mice, as well as in APOE3 and APOE4 astrocytes, provide the first evidence for the small, yet significant changes in specific signatures of cytokines and glucose metabolism due to APOE genotype, even without the presence of AD proteinopathies. Considering how APOE4 primes the brain and body for disease prior to these pathologies is essential because AD does not typically begin until after the age of 65. Thus, the AD therapeutic window may be missed if we continue to only research how APOE4 affects AD proteinopathies that likely do not begin developing until the 5th or 6th decade of life. While gaps in knowledge about the mechanisms of these changes in immunometabolism remain, my findings lay the groundwork for understanding APOE4 repercussions early in and throughout AD. It is my hope that these results will help enable greater actions towards personalized medicine in AD, encouraging APOE-specific therapeutic targets to decrease the number of people affected by this devastating disease.
If You Would Like to Learn More About My Thesis:
You can find out more by reading my published papers below. Additionally, I would be more than happy to talk with you about my findings! Please email me at rfleeman95@gmail.com if you are interested in chatting.
Fleeman RM, Snyder AM, Kuhn MK, Chan DC, Smith GC, Arnold AC, Crowley NA, Proctor EA. Predictive link between systemic metabolism and cytokine signatures in the brain of apolipoprotein E ε4 mice. Neurobiology of Aging. 2022 Dec 5; 123:154-169. doi: 10.1016/j.neurobiol aging.2022.11.015
Fleeman RM, Kuhn MK, Chan DC, Proctor EA. Apolipoprotein E ε4 modulates astrocyte neuronal support functions in the presence of amyloid-β. Journal of Neurochemistry. 2023 Feb 10. doi: 10.1111/jnc.15781
Fleeman RM, Proctor EA. Astrocytic Propagation of Tau in the Context of Alzheimer’s Disease. Frontiers in Cellular Neuroscience. 2021 Mar 17; 15:645233. doi: 10.3389/fncel.2021. 645233
If You Would Like to Learn More About Alzheimer’s Disease:
The Alzheimer’s Association Website is a fantastic resource for learning more about what Alzheimer’s disease is, how it is diagnosed, and what to do if you or a loved one experience symptoms.
TL:DR
- APOE4 is the greatest genetic risk factor for Alzheimer’s disease yet we don’t fully understand all mechanisms of risk
- APOE4 leads to immunometabolic dysregulation in the brain and body, even in the absence of Alzheimer’s disease proteinopathies
References
1. Alzheimer’s Association. 2022 Alzheimer’s disease facts and figures. Alzheimers Dement 18, 700–789 (2022).
2. van Dyck, C. H. et al. Lecanemab in Early Alzheimer’s Disease. N Engl J Med (2022) doi:10.1056/NEJMOA2212948/SUPPL_FILE/NEJMOA2212948_APPENDIX.PDF.
3. J, S. et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature 537, 50–56 (2016).
4. Budd Haeberlein, S. et al. Two Randomized Phase 3 Studies of Aducanumab in Early Alzheimer’s Disease. Journal of Prevention of Alzheimer’s Disease 9, 197–210 (2022).
5. Musiek, E. S., Gomez-Isla, T. & Holtzman, D. M. Aducanumab for Alzheimer disease: the amyloid hypothesis moves from bench to bedside. Journal of Clinical Investigation 131, (2021).
6. Martens, Y. A. et al. ApoE Cascade Hypothesis in the pathogenesis of Alzheimer’s disease and related dementias. Neuron (2022) doi:10.1016/J.NEURON.2022.03.004.
7. Corder, E. H. et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 261, 921–3 (1993).
8. Belloy, M. E., Napolioni, V. & Greicius, M. D. A Quarter Century of APOE and Alzheimer’s Disease: Progress to Date and the Path Forward. Neuron 101, 820–838 (2019).
9. Mahley, R. W. Apolipoprotein E: from cardiovascular disease to neurodegenerative disorders. (2016) doi:10.1007/s00109-016-1427-y.
10. Farrer, L. A. et al. Effects of Age, Sex, and Ethnicity on the Association Between Apolipoprotein E Genotype and Alzheimer Disease: A Meta-analysis. JAMA 278, 1349–1356 (1997).
11. Strittmatter, W. J. et al. Apolipoprotein E: high-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease. Proc Natl Acad Sci U S A 90, 1977–81 (1993).
12. Huang, Y. W. A., Zhou, B., Wernig, M. & Südhof, T. C. ApoE2, ApoE3, and ApoE4 Differentially Stimulate APP Transcription and Aβ Secretion. Cell 168, 427-441.e21 (2017).
13. Strittmatter, W. J. et al. Isoform-specific interactions of apolipoprotein E with microtubule-associated protein tau: implications for Alzheimer disease. Proc Natl Acad Sci U S A 91, 11183–6 (1994).
14. Shi, Y. et al. ApoE4 markedly exacerbates tau-mediated neurodegeneration in a mouse model of tauopathy. Nature 549, 523–527 (2017).
15. Qi, G. et al. ApoE4 Impairs Neuron-Astrocyte Coupling of Fatty Acid Metabolism. Cell Rep 34, (2021).
16. Lin, Y. T. et al. APOE4 Causes Widespread Molecular and Cellular Alterations Associated with Alzheimer’s Disease Phenotypes in Human iPSC-Derived Brain Cell Types. Neuron 98, 1141–1154 (2018).
17. Sienski, G. et al. APOE4 disrupts intracellular lipid homeostasis in human iPSC-derived glia. Sci Transl Med 13, (2021).
18. Leeuw, S. M. de et al. APOE2, E3, and E4 differentially modulate cellular homeostasis, cholesterol metabolism, and inflammatory response in isogenic iPSC-derived astrocytes. Stem Cell Reports 0, (2021).
19. Heeren, J. et al. Impaired recycling of apolipoprotein E4 is associated with intracellular cholesterol accumulation. J Biol Chem 279, 55483–92 (2004).
20. Strittmatter, W. J. & Roses, A. D. Apolipoprotein E and Alzheimer disease. Proceedings of the National Academy of Sciences 92, 4725–4727 (1995).
21. Tolar, M., Abushakra, S. & Sabbagh, M. The path forward in Alzheimer’s disease therapeutics: Reevaluating the amyloid cascade hypothesis. Alzheimer’s & Dementia (2019) doi:10.1016/j.jalz.2019.09.075.
22. Fleeman, R. M. et al. Predictive link between systemic metabolism and cytokine signatures in the brain of apolipoprotein E ε4 mice. Neurobiol Aging (2022) doi:10.1016/j.neurobiolaging.2022.11.015.
23. Fleeman, R. M., Kuhn, M. K., Chan, D. C. & Proctor, E. A. Apolipoprotein E ε4 modulates astrocyte neuronal support functions in the presence of amyloid-β. J Neurochem (2023) doi:10.1111/JNC.15781.
24. Fleeman, R. M. & Proctor, E. A. Astrocytic Propagation of Tau in the Context of Alzheimer’s Disease. Front Cell Neurosci 15, 63 (2021).
Pingback: Get on the write track for an LTS blog post | Lions Talk Science