By Rebecca Fleeman
Why are some diseases easy to diagnose and others aren’t? For instance, we can get a COVID rapid test and have a result in hours, whereas a disease like Alzheimer’s requires numerous tests over several months to make a diagnosis. Simply put, COVID is an infectious disease, caused by one single known virus, which has an easy-to-target viral protein present in your spit and nasal passages when infected with it1. However, Alzheimer’s disease is a multifaceted disease with an unknown cause and unique progression of symptoms and pathology for each afflicted person. Without knowing what causes Alzheimer’s disease, we turn to measuring pathology and symptoms for diagnosis.
Alzheimer’s disease, a neurodegenerative disease resulting in the loss of cognitive function and memory, affects over 6.2 million Americans2. Alzheimer’s has a myriad of measurable pathologies, however, being a disease of the brain, directly measuring pathology implicates a brain biopsy, which is not indicated because Alzheimer’s disease is not surgically indicated3. It is important that we diagnose Alzheimer’s as early as possible because it increases someone’s chance of benefiting from available drugs and provides people more opportunities to partake in clinical trials that may provide clinical benefit. Four drugs for Alzheimer’s disease have been on the market for close to 20 years (Donepezil, Rivastigmine, Galantamine, and Memantine), but these drugs only treat symptoms4. Unfortunately, we do not currently have any reliable therapeutics that can stop or reverse the progression of the disease5, thus participation in clinical trials is our best chance to slow the effects of Alzheimer’s disease. The lack of successful Alzheimer’s disease drugs, despite thousands of clinical trials, may be due to treating patients too late in the disease progression. Thus, early diagnosis of Alzheimer’s disease will enable the discovery of better therapeutics to treat and prevent Alzheimer’s dementia6. In order to reliably diagnose Alzheimer’s disease early in its progression, better biomarkers–measurable biological signs of disease–are needed (Figure 1).
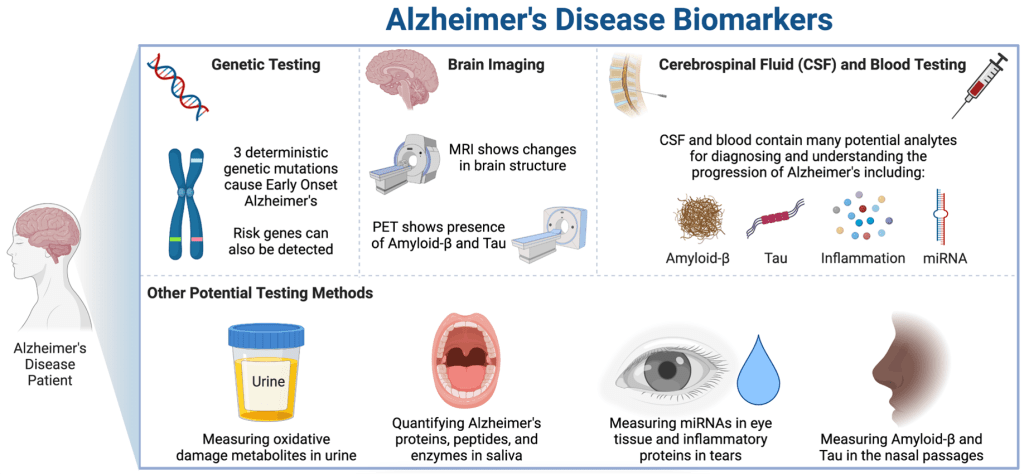
Figure made by Rebecca Fleeman in BioRender.
Genetic Testing
Alzheimer’s disease comes in two forms: early-onset (less than 5% of all cases and develops as early as mid 30s)7 and late-onset (95% of all cases and typically develops after the age of 65)5. Early-onset Alzheimer’s is heavily influenced by the genetic cards you were dealt. If someone in your family developed Alzheimer’s early on, it is recommended that you be tested for one of the three deterministic early-onset Alzheimer’s gene mutations (amyloid precursor protein, presenilin 1, and presenilin 2)7. If you carry any of these mutations, there is currently little that can be done to prevent development of the disease. For late-onset Alzheimer’s, there is not an obvious gene mutation causing the disease. Instead, we can only rely on our knowledge of risk factors to hypothesize who will develop Alzheimer’s. While you can undergo genetic testing for risk genes associated with late-onset Alzheimer’s disease (such as APOE4 testing found in 23andMe kits), these are not recommended because of the lack of prevention modalities to employ if someone carries Alzheimer’s risk genes8,9. However, scientists are hard at work studying the prevention of Alzheimer’s and how different risk factors, combined with lifestyle and environmental cues, impact Alzheimer’s risk. Thus, we may see support for preventative genetic testing in the near future.
Brain Imaging
The two hallmarks of Alzheimer’s pathology are neurofibrillary tau tangles and amyloid-β plaques in the brain3,5. These two pathological proteins can be found in the brain decades before the onset of symptoms5. The tau and amyloid-β, which typically build up in the entorhinal cortex of the brain first, lead to neuron cell death, and eventually brain atrophy. The key imaging modalities of PET and MRI aim to quantify the pathological proteins and brain atrophy, respectively. So why can’t we use brain imaging to screen for Alzheimer’s disease starting at mid-life, similar to how we screen for breast cancer annually for women after reaching 40? While amyloid-β, tau, and brain atrophy are key to an Alzheimer’s disease diagnosis, they are not completely unique to the disease. As we age, many people without Alzheimer’s disease will develop some of each of these pathologies10, and clinical evidence has shown that the quantity of amyloid-β in the brain do not always correlate with disease progression4,11. Thus, imaging cannot stand alone as a biomarker but instead must be accompanied by additional measurements. Further, a key drawback to these imaging modalities is that because they are not conclusive diagnoses, most insurances will not cover them, leaving patients to pay thousands out-of-pocket for imaging screens. Because of their expense, not every medical center even has the necessary tools or doctors for these imaging modalities, increasing health disparities.
CSF Testing
Cerebrospinal fluid (CSF) is the liquid that bathes your brain and spinal cord. Because it is in direct contact with your brain, CSF is the closet we can get to testing the pathological proteins in the brain without performing a brain biopsy12. While having high accuracy, CSF collection requires a spinal tap, an extremely painful and invasive procedure which requires highly trained physicians to perform3. Due to rapid advances in multiomics technologies over the past few years, we can assay large numbers of RNA, proteins, and metabolites from small CSF samples, enabling us to study thousands of analytes in each patient sample3. At present, the focus of CSF testing mainly involves quantifying amyloid-β and tau, as well as enzymes involved in their creation, detecting vesicular transport protein changes involved in disease progression, and profiling inflammatory signaling molecules to better understand disease state3. A newer and exciting protein that holds promise for diagnosing and monitoring the progression of Alzheimer’s is neurofilament light chain (NfL), which is upregulated in disease 3,5,12,13. Finally, brain-associated micro-RNAs (miRNAs) are a promising target of CSF studies3, as their role in regulating gene expression implicates them in Alzheimer’s disease. Of note, due to the invasive nature of CSF testing, it is difficult to run clinical trials comparing AD patients to healthy patients, because most healthy individuals do not want to undergo spinal tap. Thus, many CSF studies focus on Alzheimer’s progression characteristics (that do not require non-demented controls) so that we can more accurately determine how quickly someone’s individual disease will progress.
Blood Testing
Blood testing is highly accessible, cheaper, and less painful than CSF testing, yet does come with its own complications for Alzheimer’s biomarker testing. The complex matrix of blood containing many cells and molecules makes it more difficult than CSF to analyze3 and due to the blood-brain barrier blocking transmission of many proteins to and from the brain to periphery, many analytes do not have translatable quantities between the brain and blood. That is to say, that even if we detect X amount of a pathological protein in the blood, it may not equal the amount in the brain, thus providing a futile quantification. However, because of the simplicity of obtaining blood, progress in finding blood biomarkers is paramount. With the scientific advancement of highly sensitive assays, we are now able to quantify Alzheimer’s proteins in the blood, including amyloid-β and tau, albeit to a lower accuracy than when quantified in CSF12,14. NfL, also found in CSF, has recently been found to be highly accurate in blood plasma samples for determining progression of Alzheimer’s disease5,12,14.
Additionally, a newer avenue for blood biomarkers is measuring inflammation through platelet, neutrophil, and lymphocyte counts, as well as cytokine concentrations14,15. Inflammation of one brain cell type in particular, astrocytes, increases production of the protein glial fibrillary acidic protein (GFAP), and measuring GFAP levels in the blood was approved as a traumatic brain injury biomarker in 2018. This approval for GFAP as a biomarker for the inflammation caused in traumatic brain injury paves the way for using GFAP for other inflammatory brain diseases, such as Alzheimer’s disease12. Other promising leads for Alzheimer’s biomarkers in blood samples include studying brain-secreted extracellular vesicles (BEVs) in the blood, as well as miRNAs, similar to those seen in the CSF3.
Future Testing Methods
There are a number of unique methods for Alzheimer’s disease biomarker detection on the horizon. With an urgent need to diagnose and measure Alzheimer’s pathology non-invasively, scientists have turned to testing urine, saliva, eye tissue and tears, and also olfactory function3,16. These forward-thinking methods of detection are gaining traction and may be added to the battery of tests we currently use for Alzheimer’s testing in the near future.
While not a simple disease to diagnose or predict the progression of, Alzheimer’s disease continues to progress in the number of people it affects. By the year 2050, over 12.7 million Americans are projected to develop Alzheimer’s disease2. Scientists are hard at work to discover the best tools to diagnose early and predict the progression of disease more accurately so that better prevention strategies and therapeutics can be prescribed. Many biomarker tests are on the horizon, and as of 2021, there were 126 new Alzheimer’s disease therapies in clinical trials, 28 of which were in phase III, many of which target the underlying pathology17. Thus, early diagnosis from new biomarkers will soon be extremely meaningful as successful disease-modifying drugs for Alzheimer’s disease gain FDA approval, leading the way to a world without Alzheimer’s disease.
Want to learn more? Here are some great resources with more information on Alzheimer’s Disease:
- General Alzheimer’s information: https://www.alz.org/
- Alzheimer’s disease biomarkers: https://www.alz.org/alzheimers-dementia/research_progress/earlier-diagnosis
- Warning signs of Alzheimer’s disease and what to do if someone you know has been diagnosed: https://www.alz.org/help-support
- To get involved in Alzheimer’s disease clinical trials: https://www.alz.org/alzheimers-dementia/research_progress/clinical-trials/about-clinical-trials
TL:DR
- Alzheimer’s disease is difficult to diagnose and predict progression
- Novel Alzheimer’s biomarkers from genetic testing, brain imaging, CSF, blood, urine, saliva, and eye and olfactory function are on the rise
References
1. COVID-19 Test Basics. FDA Available at: https://www.fda.gov/consumers/consumer-updates/covid-19-test-basics.
2. Alzheimer’s Association. 2021 Alzheimer’s Disease Facts and Figures. (2021).
3. Ausó, E., Gómez-Vicente, V. & Esquiva, G. Biomarkers for Alzheimer’s Disease Early Diagnosis. J. Pers. Med. 10, 1–27 (2020).
4. Mehta, D., Jackson, R., Paul, G., Shi, J. & Sabbagh, M. Why do trials for Alzheimer’s disease drugs keep failing? A discontinued drug perspective for 2010–2015. Expert Opin. Investig. Drugs 26, 735 (2017).
5. Long, J. M. & Holtzman, D. M. Alzheimer Disease: An Update on Pathobiology and Treatment Strategies. Cell 179, 312–339 (2019).
6. Se Thoe, E., Fauzi, A., Tang, Y. Q., Chamyuang, S. & Chia, A. Y. Y. A review on advances of treatment modalities for Alzheimer’s disease. Life Sci. 276, 119129 (2021).
7. Alzheimer’s Disease Genetics Fact Sheet. National Institute on Aging (2019). Available at: https://www.nia.nih.gov/health/alzheimers-disease-genetics-fact-sheet#:~:text=
8. How Biomarkers Help Diagnose Dementia. NIH National Institute on Aging (NIA) (2022). Available at: https://www.nia.nih.gov/health/how-biomarkers-help-diagnose-dementia.
9. Genetic testing and Alzheimer’s disease. Alzheimer Society of Canada (2018). Available at: https://alzheimer.ca/en/about-dementia/what-alzheimers-disease/genetic-testing-alzheimers-disease.
10. Weber, C. J. et al. The Worldwide Alzheimer’s Disease Neuroimaging Initiative: ADNI‐3 updates and global perspectives. Alzheimer’s Dement. Transl. Res. Clin. Interv. 7, (2021).
11. Nelson, P. T. et al. Correlation of Alzheimer Disease Neuropathologic Changes With Cognitive Status: A Review of the Literature. J. Neuropathol. Exp. Neurol. 71, 362 (2012).
12. Abdelhak, A. et al. Blood GFAP as an emerging biomarker in brain and spinal cord disorders. Nat. Rev. Neurol. 2022 183 18, 158–172 (2022).
13. Lim, B. et al. CSF neurofilament light may predict progression from amnestic mild cognitive impairment to Alzheimer’s disease dementia. Neurobiol. Aging 107, 78–85 (2021).
14. Dong, X., Nao, J., Shi, J. & Zheng, D. Predictive Value of Routine Peripheral Blood Biomarkers in Alzheimer’s Disease. Front. Aging Neurosci. 11, 332 (2019).
15. Swardfager, W. et al. A Meta-Analysis of Cytokines in Alzheimer’s Disease. Biol. Psychiatry 68, 930–941 (2010).
16. Lee, J. C., Kim, S. J., Hong, S. & Kim, Y. S. Diagnosis of Alzheimer’s disease utilizing amyloid and tau as fluid biomarkers. Exp. Mol. Med. 2019 515 51, 1–10 (2019).
17. Cummings, J., Lee, G., Zhong, K., Fonseca, J. & Taghva, K. Alzheimer’s disease drug development pipeline: 2021. Alzheimer’s Dement. Transl. Res. Clin. Interv. 7, e12179 (2021).
Pingback: APOE4-get About It: How the Greatest Genetic Risk Factor for Alzheimer’s Disease affects the Brain and Body | Lions Talk Science