By Chris Kendra
Fighting the spread of Human Immunodeficiency Virus (HIV) has become easier than ever with newly developed therapeutics. In addition to current once-daily pills, new tools in the fight against the HIV epidemic have come in the form of two types of long-lasting injectable HIV medications, one as a first line prophylactic and the second as a form of HIV treatment.
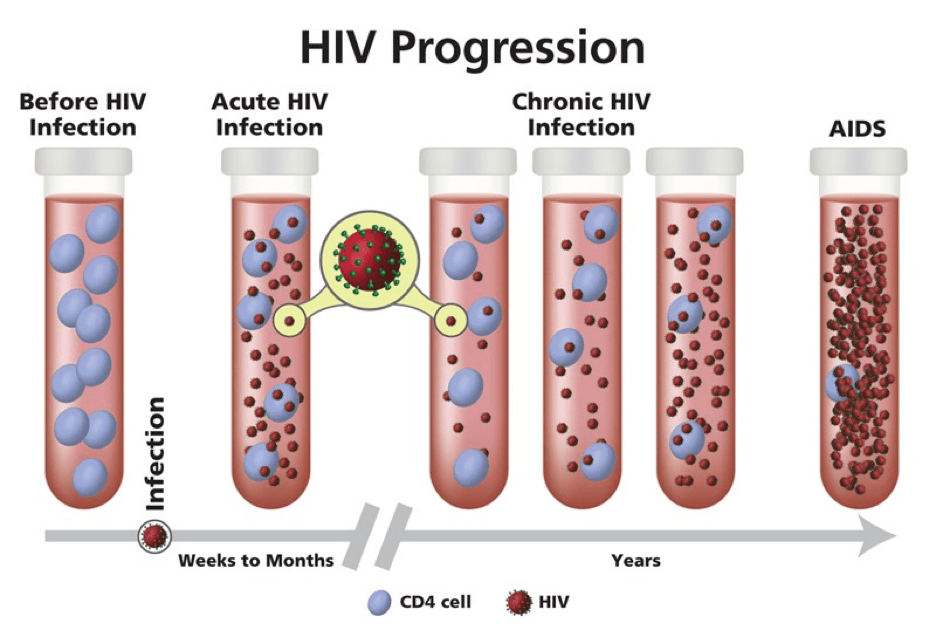
Human Immunodeficiency Virus (HIV-1 and HIV-2) is a species of lentivirus which falls within the Retroviridae family 1. As a retrovirus, HIV carries a single-stranded RNA genome within its capsid. HIV infection begins when someone is exposed to infected blood, semen, or vaginal secretions. Once in the body, HIV targets and enters CD4+ immune cells, where the HIV capsid disassembles to reveal its genome, which is then reverse transcribed into DNA and integrated into the host genome 1. From there, a lengthy infection process occurs where vast numbers of an individual’s immune cells are infected (Fig 1.). Over time, reductions in immune cell populations result in an immunocompromised state, known as acquired immunodeficiency syndrome (AIDS) 2. While the infection process follows a uniform pattern in most individuals, the timeline progression to AIDS is very heterogeneous. Some HIV infections can last up to 10 years before symptoms of AIDS begin to develop. AIDS patients are incredibly susceptible to a wide variety of infections, which are often the cause of premature mortality in these populations rather than AIDS itself. To date, there are more than 1.2 million people living with HIV in the U.S. and over 37 million people world-wide have died from HIV/AIDS3.
HIV rose to infamy in 1981 when a report from the CDC covered the death of 5 otherwise healthy young gay men in Los Angeles from Pneumocystis carinii pneumonia4,5. As the year went on, additional reports across the United States of seemingly healthy individuals succumbing to infections from opportunistic pathogens and cases of Kaposi sarcoma, a rare cancer only usually arising in the elderly and immunocompromised, arose 4,6. At the time, research indicated that the immune systems of these individuals were compromised, and most cases were occurring in persons who were gay. For this reason, the term GRID, or Gay-Related Immune Deficiency was used 7 until 1983, when the CDC officially documented the syndrome as AIDS. Cases were rising rapidly, there was still no known cause of the syndrome, and due to the nature of the disease, diagnostic tests only revealed a problem with white-cell count after it was too late to help the patient.
It was not until April 23rd of 1984 that the retrovirus Human T-lymphotropic Virus-III (HTLV-III), was determined to be the causative agent of AIDS, as reported by researchers at the National Cancer Institute8,9. From then on, tests for HTLV-III (later renamed HIV) and research into vaccines, treatment, and prevention were made a priority in the fight against this epidemic. While no cure for HIV currently exists, advances in the past 10 years of biomedical research have provided the world with new routes of disease prevention and management.
Since 2015, cases of new infections have dropped over 9% to 35,000 new cases per year and continue to drop with the addition of widely available prevention strategies and increased comprehensive sexual-health education. When it comes to prevention, those who are at high risk for HIV infection can choose to take pre-exposure prophylaxis (PrEP), a medication that helps prevent viral transmission. The most common forms of PrEP are two options of daily pills known as Truvada and Descovy that can reduce the chance of HIV transmission by over 99% when consistently taken daily10,11. Both compounds are inhibitors of HIV reverse transcriptase (Fig. 2.3), preventing the conversion of HIV RNA genome into DNA, thereby stopping infection.
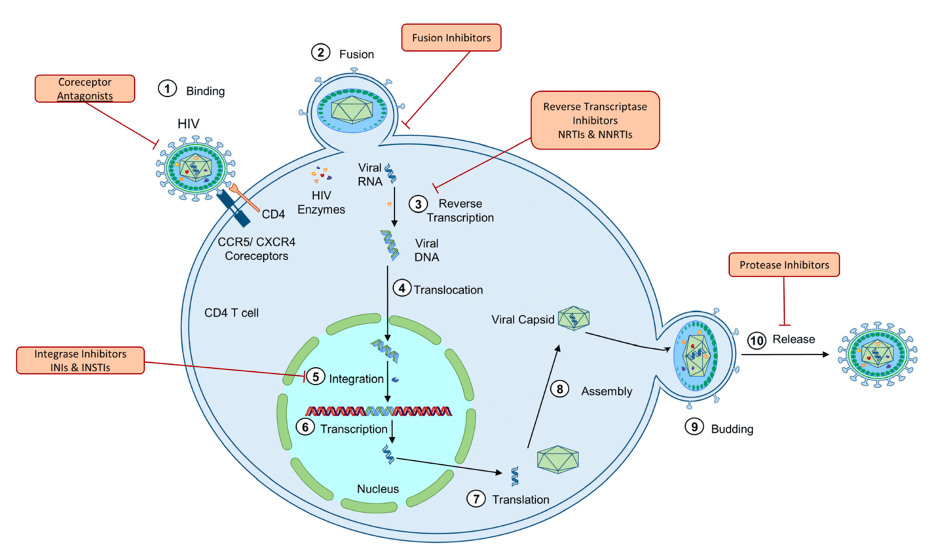
In December of 2021, an injectable therapy known as Apretude was approved by the FDA for use as a bi-monthly form of PrEP 12. Apretude is an extended-release formulation of cabotegravir, an integrase inhibitor (Fig. 2.5), preventing the integration of the HIV DNA genome into infected immune cells13. Apretude represents the first type of long-acting PrEP, which is poised to have a positive effect on individuals who are unable to conform to daily medication or who do not have the necessary resources or access to frequent prescription refills.
While PrEP offers great protection for those trying to prevent HIV infection, treatments for individuals with HIV have also come very far. Currently, treatments for HIV infection focuses on preventing the disease progression to AIDS by treating an individual with antiretroviral therapy (ART) until their infection is considered undetectable. ART combines multiple agents taken as daily medication that inhibit the reverse transcription and integration of the virus (Fig. 2.3, 2.5), prevent new viral particles from forming (Fig. 2.8, 2.9, 2.10), and prevent HIV from entering and infecting new immune cells (Fig. 2.1, 2.2)14. In January of 2021, the FDA approved an injectable form of HIV ART known as Cabenuva15. This once-monthly injection combines an integrase inhibitor and reverse transcriptase inhibitor to treat HIV-1 infection and helps patients achieve and maintain undetectable viral loads. In comparison to the usual ART regimen requiring daily oral doses of medication, this advancement makes the management of HIV much more manageable and ensures that those with HIV can live with suppressed levels of virus in their system. Prior to ART, HIV infection was considered a terminal illness with significantly reduced life expectancy due to the complications from AIDS. However, while undergoing ART, HIV positive individuals with undetectable levels of the virus are unable to transmit HIV to anyone else and live full lives to ages comparable of those without HIV16,17.
With the addition of these new choices in HIV PrEP and ART, preventing HIV infection and stopping the spread of HIV is one step closer to ending the AIDS epidemic. However, the first step for an individual to help stop the spread of HIV is to get tested and know their own HIV status. Only together can we beat HIV.
TL: DR
- There are >1.2 million cases of HIV in the US, with over 35,000 new infections annually
- Two long-lasting injectable therapies are now offered for HIV prevention and treatment
- Stopping the spread of HIV starts with getting tested regularly and knowing your HIV status
References
1. Clinical Information Human Immunodeficiency Virus (HIV). 2016;doi:10.1159/000445852
2. Aids IoMCoaNSf. Clinical Manifestations of HIV Infection. 1986;
3. Global HIV & AIDS statistics — Fact sheet | UNAIDS.
4. Morbidity and mortality weekly report, Vol. 30, no. 25, July 3, 1981. 1981.
5. Pneumocystis Pneumonia — Los Angeles.
6. Cesarman E, Damania B, Krown SE, Martin J, Bower M, Whitby D. Kaposi Sarcoma. Nature reviews Disease primers. 2019/12// 2019;5(1):9-9. doi:10.1038/S41572-019-0060-9
7. NEW HOMOSEXUAL DISORDER WORRIES HEALTH OFFICIALS – The New York Times.
8. Broder S, Gallo RC. A pathogenic retrovirus (HTLV-III) linked to AIDS. The New England journal of medicine. 1984/11// 1984;311(20):1292-1297. doi:10.1056/NEJM198411153112006
9. Gallo RC, Montagnier L. The Discovery of HIV as the Cause of AIDS. http://dxdoiorg/101056/NEJMp038194. 2009/10// 2009;349(24):2283-2285. doi:10.1056/NEJMP038194
10. U.S. Food and Drug Administration Approves Gilead’s Truvada® for Reducing the Risk of Acquiring HIV. https://www.gilead.com/news-and-press/press-room/press-releases/2012/7/us-food-and-drug-administration-approves-gileads-truvada-for-reducing-the-risk-of-acquiring-hiv
11. FDA approves second drug to prevent HIV infection as part of ongoing efforts to end the HIV epidemic. https://www.fda.gov/news-events/press-announcements/fda-approves-second-drug-prevent-hiv-infection-part-ongoing-efforts-end-hiv-epidemic
12. FDA Approves First Injectable Treatment for HIV Pre-Exposure Prevention. https://www.fda.gov/news-events/press-announcements/fda-approves-first-injectable-treatment-hiv-pre-exposure-prevention
13. APRETUDE (cabotegravir extended-release injectable suspension), for intramuscular use. https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Apretude/pdf/APRETUDE-PI-PIL-IFU.PDF
14. Saag MS, Gandhi RT, Hoy JF, et al. Antiretroviral Drugs for Treatment and Prevention of HIV Infection in Adults: 2020 Recommendations of the International Antiviral Society-USA Panel. JAMA. 10 27 2020;324(16):1651-1669. doi:10.1001/jama.2020.17025 15. FDA. Drug Trials Snapshot: CABENUVA. https://www.fda.gov/drugs/drug-approvals-and-databases/drug-trials-snapshot-cabenuva
Hi,
Thanks for sharing this wonderful article. It is very good information about some the curing of HIV/AIDS infection. It was very useful for many, including me to explore and gather the information about HIV/AIDS. It is a must-read blog to explore the new information about HIV infection curable medicines.Keep posting
Thanks for detailing different ways to deal with an HIV infection. It’s good to know that fighting it has become easier than ever with newly developed methods. I think it’s great that there are now so many options to help people with HIV.