By Victoria Silvis
While the world has focused on rapidly developing a vaccine for SARS-CoV2, the WHO recently approved a vaccine that has been developed for malaria, a serious illness endemic to poorer tropic and subtropic regions globally. In 2020 alone, there were 229 million cases with an estimated half a million deaths.1 Malaria infections occur regardless of age or gender, but in children and pregnant people, infection can quickly become fatal. Although there have been great strides in reducing the mortality rate from 25 in 2000 to 10 in 2019, malaria is still a serious threat to individuals living in endemic regions (Figure 1)1. Sub-Saharan Africa, regions of South-East Asia, the Eastern Mediterranean, Western Pacific, and the Americas are all areas of high transmission, with sub-Saharan Africa having the highest number of cases and deaths.2

First, let’s establish what causes malaria. Malaria is caused by a parasite Plasmodium falciparum. There are other species in the Plasmodium family: P. vivax, P. ovale, and P. malariae, but P. falciparum is the most common and therefore will be the focus here. P. falciparum is carried by female Anopheles mosquitos after biting an infected person and is then transmitted to the next person the mosquito bites, making the mosquito a vector. The hatred of a mosquito bite in the US comes with an irritating itch but doesn’t come with the fear of malaria transmission like it does in endemic countries as it is considered eliminated in the US. A mosquito bite is the typical route of transmission, however blood transfusion, organ transplant, needle sharing, and mothers transmitting to children during delivery are alternative transmission routes.4
Malaria can be categorized as uncomplicated or severe. Uncomplicated infection feels like the flu, with symptoms including fever, chills, sweats, nausea, and body aches. Often in countries with infrequent malaria cases, uncomplicated infection can go undiagnosed or misdiagnosed due to the symptom similarities with more common infections. Severe malaria occurs when organ failure or blood abnormalities are exhibited and can manifest in a variety of ways including cerebral complications severe anemia, acute kidney injury, metabolic acidosis, and acute respiratory distress. Uncomplicated malaria is often self-treated in endemic regions; however, severe malaria is a medical emergency and requires immediate and aggressive treatment.5
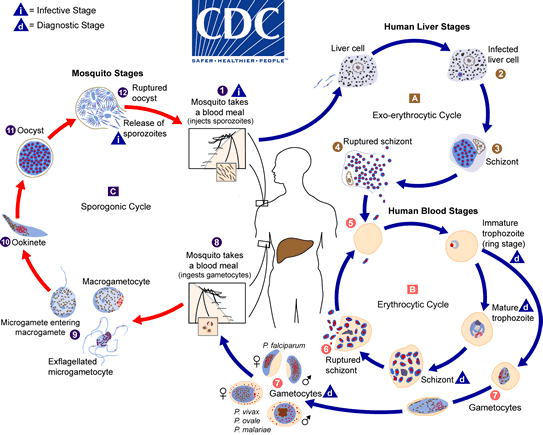
P. falciparum has multiple lifecycles but there are a few important steps to highlight (Figure 2). There is a sporogonic cycle (C) of replication within the mosquito where P. falciparum gametocytes (7) multiply to form sporozoites (12), which upon human inoculation infect the liver (A). The sporozoites mature within the liver forming a schizont (3) before rupturing and infecting red blood cells (5) which is when symptoms start to appear. The lifecycle in the bloodstream, or erythrocytic cycle (B), will prolong the infection in the individual while forming gametocytes (7) which allow replication within the next hungry mosquito perpetuating the transmission of malaria to others4.
Those who plan to travel to malaria endemic regions should inform their doctor to obtain a prophylactic prescription for antimalarial medications. Additionally, keeping mosquitos off you by any means necessary, such as the commonly used insecticide-treated bed nets, is incredibly important in preventing malaria6.
Now you may ask, why do we need a vaccine when malaria is treatable and preventable with prescription drugs? Unfortunately, most of the hot-spot transmission regions have insufficient resources or poor socio-economic stability causing access to preventative measures, like insecticides and bed nets, to be slim or non-existent in some cases7. Additionally, eradication efforts including insecticides and drugs have limited participation and instead have led to drug and insecticide resistance8. The Malaria Consortium is one of the leading non-profit organizations working to control and treat malaria and other neglected tropical diseases9. The Malaria Consortium provides seasonal malaria chemoprevention (SMC), which is a combination of two antimalarial drugs: sulfadoxine-pyrimethamine (SP) and amodiaquine (AQ), to children under three. SMC has been shown to prevent 75% of malaria cases in children under five in clinical trials9. While incredibly beneficial, widespread SMC provision requires community participation, volunteer distributors, drug procurement, and funding10. The best-case scenario is that malaria could be prevented from the get-go.
That’s where the RTS,S/AS01E (Mosquirix) recombinant malaria vaccine comes in. Mosquirix was developed by GSK and the PATH Malaria Vaccine Initiative and has reduced childhood malaria cases by 39% in just 4 years.11 With the success in phase 3 trials, the WHO advocated for a pilot program to be rolled out in Ghana, Kenya, and Malawi in 2019. The Mosquirix pilot has reached an estimated 800,000 children already and will continue through 2023. Mosquirix is offered along with other childhood vaccines and as such, families with access to routine immunizations, also have access to Mosquirix.12
SMC has been the gold-standard of malaria prevention, so how effective is Mosquirix compared to SMC? The results of a trial performed in Burkina Faso and Mali were recently published in NEJM comparing Mosquirix, SMC, and the combination of both interventions. The trial began in 2017 with 6861 children between age 5 and 17 months of age randomly assigned to vaccine-alone, chemoprevention-alone, or combination groups. Children in vaccination groups received five total doses, with the first three each one month apart, and the final two each one year from the last administration. Children in chemoprevention groups received SP and AQ monthly for four courses each year, beginning in August of year 1, before the high transmission season starting in December. Participants received placebos of either vaccine or chemoprevention based on group inclusion, and all participants were given insecticide treated bed nets at time of inclusion. Interestingly, the chemoprevention alone and vaccine alone groups had similar incidence rates of 304.8 and 278.2 events per 1000 person-years at risk respectively. The combination group had a reduction to 113 events per 1000 person-years at risk indicating increased protective efficacy. The combined intervention also had increased protection against hospitalization and severe malaria infection.13
While combined chemoprevention and vaccination provide the best protection from malaria, having either chemoprevention or vaccination remains beneficial to children in malaria-endemic regions. These interventions will not stop malaria overnight but are an added tool to the disease prevention toolbox. Even though it doesn’t happen on our doorstep, malaria is still very real and incredibly deadly. To find out more about malaria, other tropical disease prevention, or ways to help the cause, visit the Malaria Consortium website.
TL:DR
- Malaria is still a serious threat globally, with almost half a million deaths annually.
- Interventions only work as well as community participation and socioeconomic support allow.
- A recently approved vaccine shows promise and when combined with already available chemoprevention is incredibly effective against childhood malaria infections and deaths.
References
- https://www.who.int/publications/i/item/9789240015791.
- https://www.who.int/news-room/fact-sheets/detail/malaria
- https://www.cdc.gov/malaria/about/distribution.html
- https://www.cdc.gov/malaria/about/biology/index.html
- https://www.cdc.gov/malaria/about/disease.html
- https://www.cdc.gov/malaria/about/faqs.html
- https://www.cdc.gov/malaria/malaria_worldwide/impact.html
- https://www.cdc.gov/malaria/about/history/
- https://www.malariaconsortium.org/pages/who_we_are.htm
- https://www.malariaconsortium.org/pages/seasonal-malaria-chemoprevention/our-smc-programme.htm
- RTS,S Clinical Trials Partnership. Efficacy and safety of RTS,S/AS01 malaria vaccine with or without a booster dose in infants and children in Africa: final results of a phase 3, individually randomised, controlled trial. Lancet. 2015 July 4-10;386(9988):31-45.
- https://www.who.int/news-room/q-a-detail/q-a-on-rts-s-malaria-vaccine
- Chandramohan D, Zongo I, Sagara I, Cairns M, Yerbanga R.-S., Diarra M, et al. Seasonal malaria vaccination with or without seasonal malaria chemoprevention. N Engl J Med. 2021 Aug 26;385:1005-17.
Pingback: Drink to Your Health: The Gin and Tonic Story | Lions Talk Science