By Gaelyn Lyons
There has been a lot of talk lately about the COVID-19 booster vaccine, especially with new variants arising and Pfizer submitting an application to the FDA for a booster against COVID-19. The first two jabs have been found to significantly protect individuals from severe symptoms and reduce the risk of hospitalization.1 However, due to vaccine hesitancy and vaccine refusal, the variants continue to evolve.2
You’re probably sitting there thinking, “I got both of my vaccines, shouldn’t I be fine?” And the answer is, kind of.
In early September 2021, a study was published looking at the level of antibodies that were present in healthy individuals six months after receiving their second dose of the Pfizer/BioNTech vaccine BNT162b2 (Figure 1).3 Initially, there was a significantly higher amount of antibodies against the SARS-CoV-2 protein that binds to the host cell after the second dose of BNT162b2 compared to the first dose. However, six months after the second dose, the concentration of antibodies decreased to levels similar to that of after the first dose.3
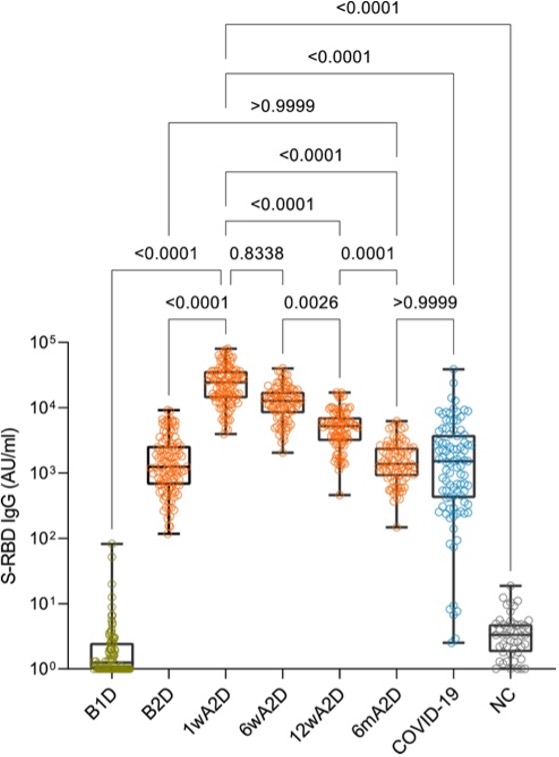
This isn’t the first time scientists, medical doctors, and public health experts have seen a decline of neutralizing antibodies after vaccination. Vaccines against pertussis (whooping cough), mumps, and influenza have all demonstrated this waning immunity.5 As children we received these routine vaccines and as adults we continue to receive their boosters to build up lasting immunity.6 Notably, just because our antibodies are decreasing doesn’t mean we aren’t protected from these diseases. Our memory B cells and T cells are able to last much longer than antibodies and are able to produce more when they are re-exposed to the virus.7 However, since COVID-19 is a novel virus, scientists are still collecting data to understand how long our immunity lasts to both the COVID-19 virus and its vaccine.
The evolving variants pose another issue that may necessitate boosters. Boosters can be tailored to target mutations present in variants to make them more effective against these variants.6 While the current vaccines are able to prevent severe symptoms, breakthrough infections and variants are on the rise due to a large number of unvaccinated individuals.2
The FDA hosted a public meeting on September 15th, 2021 that included the Israeli Ministry of Health, the University of Bristol, U.K., the CDC, public health experts, and independent scientists to gain input on whether a COVID-19 booster is necessary.8 The FDA officially made the decision that a booster dose of the Pfizer vaccine would be effective in preventing COVID-19, however, those currently approved for the boosters include individuals who are 65 and older, individuals between the ages of 18 and 64 who are at high risk of severe COVID-19, and individuals between the ages of 18 and 64 whose institution or place of occupation poses a high-risk to COVID-19 exposure.8 As of now, this decision is only applicable to the Pfizer vaccine since it has been approved for use longer than the Moderna and J&J/Jansen vaccines; however, this will change as the latter companies submit new data.
As scientists, we are familiar with the evolving changes to how we treat diseases based on data that is collected, which is known as the scientific method (Figure 2). Based on previous data and observations, we make a hypothesis.9 We then experimentally test that hypothesis, obtain more data, and then create new hypotheses based off of those results. We understand that this process is cyclical, however, the general public mainly observes the final results and conclusions that scientists come to and do not see the results that go against our original hypothesis.
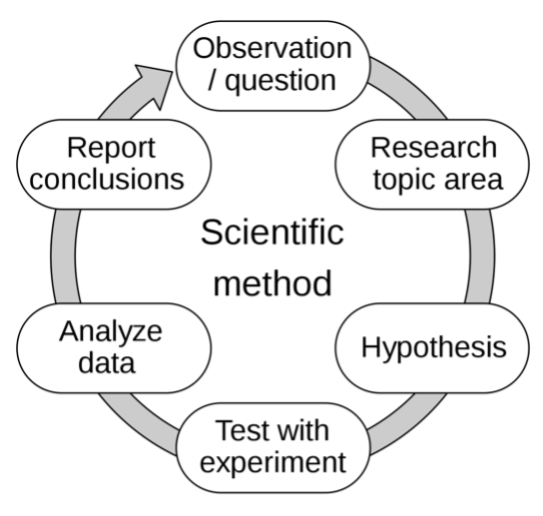
Applied to COVID-19, the hypothesis we had in the beginning of 2020 to minimize spread of COVID-19 (disinfecting surfaces and quarantining helps decrease the spread of COVID-19) looks much different than the one we are working with now (receiving a booster vaccine six months after getting the second vaccine can help build up more immunity against COVID-19). With the pandemic, the public is seeing the scientific method in action. To someone who is not used to this, it may look like scientists are constantly changing their minds and this could lead to uneasiness.
Scientists have a responsibility to help guide those who may not understand how the scientific method is working. Going through the pandemic is a new experience for all of us and the way we deal with it will change as we gather more data. In terms of whether you should get a COVID booster, the answer is that it’s up to you. If you feel like you’ll benefit from receiving a third jab, go for it! It’s also a good idea for those who the FDA recommends to receive the booster to get one. However, the most likely way we’ll decrease the evolution of the variants is to get more people vaccinated. If you do not fall under the FDA’s criteria for those who are highly recommended to get the vaccine, the most important thing you can do is to continue following the CDC guidelines of wearing masks, social distancing, and encouraging those we know who are unvaccinated to get the vaccine.
TL:DR
- A recent study identified waning immunity in individuals six months after receiving their second COVID vaccine.
- The FDA has approved the use of the Pfizer booster vaccine for select group.
References
1. Majeed, A., Papaluca, M. & Molokhia, M. Assessing the long-term safety and efficacy of COVID-19 vaccines. doi:10.1177/01410768211013437.
2. Rubin, R. COVID-19 Vaccines vs Variants—Determining How Much Immunity Is Enough. JAMA 325, 1241–1243 (2021).
3. Naaber, P. et al. Dynamics of antibody response to BNT162b2 vaccine after six months: a longitudinal prospective study. Lancet Reg. Heal. – Eur. 100208 (2021) doi:10.1016/J.LANEPE.2021.100208.
4. Charles A Janeway, J., Travers, P., Walport, M. & Shlomchik, M. J. The distribution and functions of immunoglobulin isotypes. (2001).
5. Waning Immunity Creates Risk. PharmacyTImes https://www.pharmacytimes.com/view/waning-immunity-creates-risk (2020).
6. Macmillan, C. Will You Need a COVID-19 Booster? What We Know So Far > News > Yale Medicine. Yale Medicine https://www.yalemedicine.org/news/covid-19-booster (2021).
7. Dolgin, E. COVID vaccine immunity is waning — how much does that matter? Nature 597, 606–607 (2021).
8. FDA Authorizes Booster Dose of Pfizer-BioNTech COVID-19 Vaccine for Certain Populations | FDA. https://www.fda.gov/news-events/press-announcements/fda-authorizes-booster-dose-pfizer-biontech-covid-19-vaccine-certain-populations.
9. Heldt, C. The Scientific Method on Display | Michigan Tech Research Blog. Michigan Tech https://www.mtu.edu/unscripted/2020/08/the-scientific-method-on-display.html (2020).
10. About the null and alternative hypotheses – Minitab. https://support.minitab.com/en-us/minitab/18/help-and-how-to/statistics/basic-statistics/supporting-topics/basics/null-and-alternative-hypotheses/.
This article is well written, accurate and exactly what I would expect from this author.