By Rebecca Fleeman
If you could change anything about yourself, what would it be? Would you add a couple of inches to your height to improve your view at concerts? Maybe you’d tack on some IQ points to get into the school of your choice. Or, if you read my last article, maybe you’d increase your athletic power with an ACTN3 gene mutation.
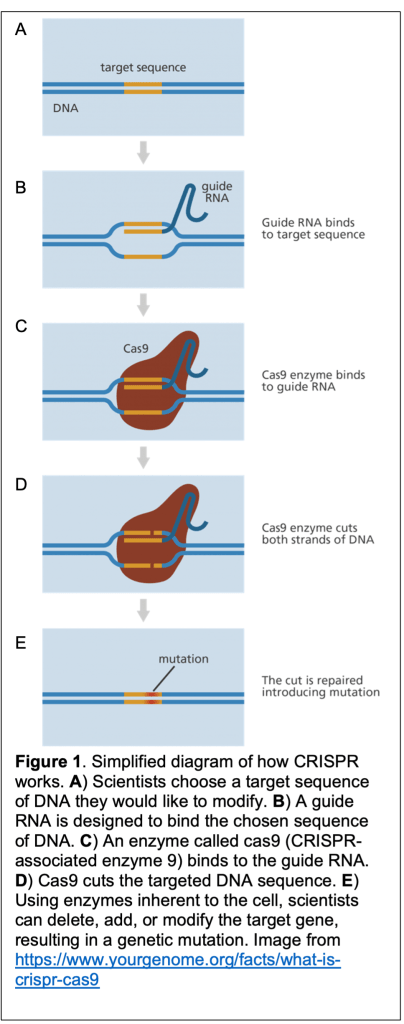
Previously, many of us had only dreamed of changing our inherited disposition, but with the technology of CRISPR, editing our genome to our liking is within grasp. CRISPR, which stands for Clustered Regularly Interspaced Short Palindromic Repeats, is a gene-editing technology developed over decades by a team of scientists, including Nobel prize-winning Jennifer Doudna and Emmanuelle Charpentier (see Figure 1 for mechanistic details of CRISPR). While other forms of gene editing predate CRISPR (TALENs and ZFNs), CRISPR is more precise, faster, easier, and importantly, cheaper.
CRISPR has come a long way from its origin over 28 years ago, beginning with the discovery of the CRISPR locus by Francisco Mojica and advancing to use in human cells by Feng Zhang1. CRISPR is now a reliable technology, climbing in prominence and currently in numerous clinical trials for blood diseases2, cancer3, blindness4, and other conditions5.
While many scientists have used CRISPR in their lab for purposes such as testing protein knockouts in cell lines and genetically altering mice to understand disease, it is easy to neglect the effects that CRISPR has on larger scale applications. From treating life-threatening diseases to editing genes of unborn babies, and beyond, it is imperative that everyone decide where they stand on the ethics of using gene editing in humans. CRISPR is an impressive technology and we, as individuals and as a society, will need to draw lines in the coming years as gene editing technology becomes more prevalent.
I recently read the #1 New York Times Best Seller, “The Code Breaker: Jennifer Doudna, Gene Editing, and the Future of the Human Race” by Walter Isaacson6. He captures the dramatic story of the race between several scientists to harness the gene cutting abilities of bacteria for biomedical exploration. On top of giving an in-depth biography of Doudna and her rise to scientific stardom, Isaacson takes the reader through a journey of ethical considerations that will undeniably shape the future of the human race. Below are some of the ethical arguments brought up in “The Code Breaker” that I thought were especially notable.
The “Playing God” Argument
This ethical debate dates back centuries, applying to the ever-evolving field of science labeling aspects of life as natural and unnatural. Many that believe God created mankind argue that each person’s genes are chosen by God and should not be under the control of man. This way of thinking elicits calling genome editing “unnatural”. The dissenting argument is that because CRISPR exploits the natural ability of bacteria to evolve and change their DNA, it is natural itself.
Using CRISPR to cure diseases is just another way humans can use their environment to protect themselves, like we use wood from trees to build a home. Others, like James Watson, one of the discoverers of the DNA helix, are yet on a third side of this argument and say that genome editing is unnatural, yet we should still take advantage of it. Watson is quoted as saying “If scientists don’t play God, who will?”6
The “This Disease Makes Me, Me” Argument
Another argument hotly debated by citizen scientists interested in genome editing is whether genetic manipulation to prevent neurological disorders is warranted. It is fiercely debated whether it is right to prevent those who will not be neurotypical from being born with their disorder. Some, like Josiah Zayner, a well-known biohacker who has used CRISPR on himself, struggles with bipolar disorder and says he has no doubt that if he could prevent having been born with bipolar, he would have, because his bipolar disorder has caused him a great deal of hardship.
Others argue that deviation from “normal” is what gives people their personality and makes them who they are. Some even argue that we could lose many of our future imaginative minds, as it is well-known that hundreds of geniuses and great artists have been neurodivergent. Albert Einstein was said to be dyslexic, Tim Burton autistic, Demi Lovato bipolar, and Vincent van Gogh schizophrenic. Without this neurodivergence, these people may have had vastly different life trajectories.
In addition to neurological disorders, it is also a point of argument to say whether abnormal developments such as blindness and deafness should be prevented. Many people who are blind or deaf challenge the belief of everyone wanting treatment or prevention for these conditions because they make the person who they are. They have grown to appreciate how their genetic predispositions shape their interactions with the world.
The “Increasing Society’s Gap in Equity” Argument
The arguments so far presented have been “what are the repercussions if we edit the genome?”. However, we may get to a point soon where we instead ask, “what are the repercussions if we don’t edit the genome?”. There may come an inflection point where so many people have genome-edited babies, that choosing not to edit your offspring could be considered immoral. Envision a future where everyone edits their babies to change risk factor genes for obesity, Alzheimer’s disease, Amyotrophic lateral sclerosis (ALS), cancer, and more. If everyone began doing this, but you didn’t, others might criticize you for not giving your child the best care possible. Inevitably we will have the issue where some do not want to edit their offspring but will do so out of social pressure.
The cost of genome editing adds to the complexity of the concept. If germline editing is not free, you can imagine that the equity gap in our society, that is already large, could expand as the rich will produce more adept children, while the poor will remain with “normal children.” Notably, CRISPR has been touted for its cheap price tag and accessibility. Thus, as the public gains the ability to buy the tools necessary to perform home-genome editing, certain regulations may need to be implemented6.
The “Obvious Line” Argument
A final perspective to consider is the “obvious line” argument, where gene editing should only be performed at the direst need, and philosophical “what ifs” are too far off to worry about. The “obvious line” is drawn where germline gene editing should only be performed for diseases that are entirely caused by simple gene mutations and will significantly affect a child’s wellbeing and/or lifespan. These cases include diseases like muscular dystrophy, Huntington’s disease, and cystic fibrosis. We know the single gene that is awry in these diseases which makes gene editing more straightforward. Removing these diseases will not give any child any “advantage”, it will simply prevent them from enduring the awful disease.
As you contemplate the arguments above, consider where, if at all, you would draw the line on using gene editing. Maybe your line is drawn at preventing risk for obesity or allergies, or maybe your line is single-gene diseases and not just risk factor diseases. Is providing your kids with a good education different than providing them with good genes? The goal of both is to set your child up to be successful.
Also consider who should make the decision. Is this something everyone should vote on in local government, or have the federal government decide, or have world organizations handle? If every state or every country had a different rule, how might this lead to imbalances in our world?
Notably, in vitro fertilization (IVF) was controversial at first, yet now it is widely accepted for parents with trouble conceiving8. The first test-tube baby was born in 1978 but now, more than 30 years later, IVF is widely accepted.
I think Walter Isaacson’s explanation of “treatment” vs “enhancement” gives the best argument for drawing the line. In this thought experiment, you consider the edit and ask if giving x edit to everyone would remove the benefit completely. For example, if you wanted to make your child taller, note that making every child taller removes the benefit of adding a few inches to your child’s height. Your child would no longer be “tall” because everyone’s height increased. Conversely, if you edited out the mutation causing Huntington’s disease in your child, and also gave this edit to every child, there would still be a benefit because already very few people in society are affected by Huntington’s. It doesn’t give someone a leg up to not have the disease, it just prevents them from the devastating disease.
Final Thoughts to Consider
Instead of genome editing before birth, should it be up to each child, later in their life when they can make the decision themselves, to choose? We could allow every 18-year-old to get their genome sequenced then decide whether they want to edit their cells. This may not be feasible for all diseases, but for many it could be. Additionally, CRISPR may not be the magic cure-all some hope it to be. Some argue that things like pre-implementation diagnostics, which are currently in use, are good enough.
While I did not mention every ethical consideration, it is evident that our society will have many CRISPR-related decisions in the near future. A wise man once said, “The people who are crazy enough to think they can change the world, are the ones who do”9. So go out and discuss the ethics of genome editing, your actions on these discussions may change the world.
Further Reading
If you would like more information on the current regulations and guidelines on genome editing, please check out the following sources:
https://www.annualreviews.org/doi/10.1146/annurev-med-112717-094629
https://ghrp.biomedcentral.com/articles/10.1186/s41256-020-00153-4
https://www.ncbi.nlm.nih.gov/books/NBK447266/
TL;DR
- CRISPR has enabled gene editing to be cheap and easy
- Where do you fall on the morals of gene editing? Important considerations:
- Is gene-editing “playing God”?
- Do diseases make someone who they are?
- Will gene editing enlarge the equity gap?
- Is there an obvious line for when to use gene editing in humans?
References
1. Broad Institute. CRISPR Timeline.
2. Frangoul, H. et al. CRISPR-Cas9 Gene Editing for Sickle Cell Disease and β-Thalassemia. https://doi.org/10.1056/NEJMoa2031054 384, 252–260 (2020).
3. Lu, Y. et al. Safety and feasibility of CRISPR-edited T cells in patients with refractory non-small-cell lung cancer. Nat. Med. 2020 265 26, 732–740 (2020).
4. First CRISPR therapy dosed. Nat. Biotechnol. 38, 382–382 (2020).
5. Gillmore, J. D. et al. CRISPR-Cas9 In Vivo Gene Editing for Transthyretin Amyloidosis. https://doi.org/10.1056/NEJMoa2107454 (2021). doi:10.1056/NEJMOA2107454
6. Isaacson, W. The Code Breaker: Jennifer Doudna, Gene Editing, and the Future of the Human Race. (Simon & Schuster, 2021).
7. Coller, B. S. Ethics of Human Genome Editing. https://doi.org/10.1146/annurev-med-112717-094629 70, 289–305 (2019).
8. Britannica, The Editors of Encyclopaedia. “In vitro fertilization”. Encyclopedia Britannica, 16 Jan. 2020, https://www.britannica.com/science/in-vitro-fertilization. Accessed 14 August 2021.
9. Steve Jobs, Apple’s “Think Different” Commercial, 1997
Pingback: De-extinction: What are the odds you’ll be eaten by a dinosaur? | Lions Talk Science