By Joseph Cirilo
Have you ever experienced one of those days where you come home after a long day of work, sit down to relax to some Netflix or Hulu, and then whatever you decide to watch throws work right back into your face? Well, I recently had this experience while watching the TV show One Tree Hill. As I started season two of the show, it was revealed that a character had a disease known as HCM, or hypertrophic cardiomyopathy (1). In the Yengo Laboratory, the lab I work in here at PSCOM, we study myosins in general, with a strong focus on how mutations in cardiac myosin can lead to HCM or its opposite yet equally detrimental counterpart, DCM (dilated cardiomyopathy). HCM is the premier cause of sudden cardiac death in young people, affecting about 1 in 200 individuals globally (2). HCM can be characterized by left ventricular hypertrophy (thickening of the ventricular wall), myocyte disarray, diastolic dysfunction, and increased contractility. Interestingly, many of the mutations causing HCM follow a dominant pattern of inheritance; that is, you only need one copy of the mutation to have the disease (3). One Tree Hill used this dominant inheritance pattern to further the plot, as the character’s two sons went to get tested since they had a “50/50” chance of having the disorder themselves (4). With the prevalence and severity of HCM well established, we are left with a fundamental question: how does a single nucleotide change in the DNA sequence lead to a complete restructuring of the heart and disease?

To understand this question, it is important to understand what cardiac myosin does within your body, as well as the protein’s structure. In humans, the gene MYH7 encodes for the β-myosin heavy chain. This protein is an actin-based ATPase motor; in other words, it uses a cycle of ATP binding and hydrolysis to create a net displacement on an actin cytoskeleton track and move forward. For cardiac myosin, the heavy chain and its two light chains, known simply as the regulatory light chain (RLC) and essential light chain (ELC), make up the “thick filament.” The actin cytoskeleton and its accessory proteins make up the “thin filament”. The two filaments together form the sarcomere, a subunit of myofibrils (muscle fibers) (5). A coordination between both of these filaments within cardiomyocytes, the cells that make up the heart muscle tissue, is responsible for proper heart muscle contraction. If we imagine the heart as a giant rowboat, the myosin would be the rowers and the actin would be the water. The rowers (myosin) need to work in sync in order for the boat (heart pumping) to properly move.
What happens when the rowers are not able to work properly in sync? What happens when one or two rowers are rowing much faster than the others, or not rowing at all? The boat is no longer able to follow that pattern and the movement on the water becomes dysregulated. The same goes for cardiac myosin pumping blood; if the ensemble is not working properly in tandem because it consists of a mixture of normal (WT) cardiac myosin and mutant myosin, dysregulation of the heart’s pumping may occur. Remember, it only takes one bad copy of the gene for this to occur. As a result, this dysregulation can lead to enhanced muscle contractility, impaired relaxation of heart tissue, and ultimately a thickening of the ventricular wall (Figure 1) (5). The question then becomes, how do mutations in cardiac myosin lead to “stronger rowers” or “slower rowers”?
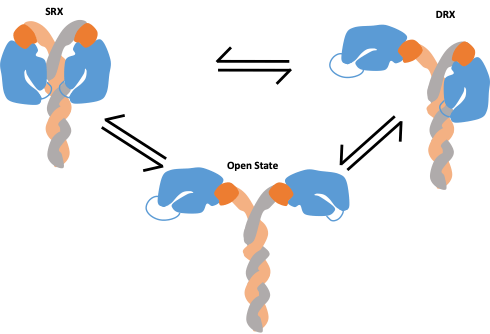
This question ultimately comes down to altered myosin regulation. One such method of regulation is the coordination between the super relaxed state (SRX) and disordered relaxed state (DRX). In SRX, both “heads” of the myosin molecule are blocked from interactions with their actin substrate, while in DRX only a single head is inhibited (Figure 2). When the amount of myosin heads that are able to interact with the actin thin filaments is altered, the overall motor properties of the myosin molecule, and thus contractility, are altered. This is especially true when a mixture of both WT myosin and mutated myosin are within a sarcomere. Interestingly, this balance between SRX and DRX is not modulated via the myosin molecule alone, but various accessory proteins that bind the myosin heavy chain can cause alterations in this process as well (6). Similarly, mutations in these proteins can also lead to HCM and altered cardiac function.
For us in science, we are taught early on how biology is just the coordination of many smaller systems building up a more complex system. From DNA and proteins to tissues and organs, everything within the system of the human body is related and can affect one another; it can take only a single DNA nucleotide change to cause a complete restructuring of an organ and a change in physiology. Though the large-scale effect that this single change makes can be surprising, I think the biggest surprise of all is that an early 2000s teen drama not only properly discussed a complex disease, but also held up 15 years later.
Citations
1. Prange G. One Tree Hill. The Desperate Kingdom of Love2004. p. 42 min.
2. Semsarian C, Ingles J, Maron MS, Maron BJ. New perspectives on the prevalence of hypertrophic cardiomyopathy. J Am Coll Cardiol. 2015;65(12):1249-54. Epub 2015/03/31. doi: 10.1016/j.jacc.2015.01.019. PubMed PMID: 25814232.
3. Gunther LK, Rohde JA, Tang W, Walton SD, Unrath WC, Trivedi DV, Muretta JM, Thomas DD, Yengo CM. Converter domain mutations in myosin alter structural kinetics and motor function. The Journal of biological chemistry. 2019;294(5):1554-67. Epub 2018/12/05. doi: 10.1074/jbc.RA118.006128. PubMed PMID: 30518549.
4. Dickson B. One Tree Hill. Truth, Bitter Truth2004. p. 42 min.
5. Muresan ID, Agoston-Coldea L. Phenotypes of hypertrophic cardiomyopathy: genetics, clinics, and modular imaging. Heart Failure Reviews. 2020. doi: 10.1007/s10741-020-09931-1.
6. Toepfer CN, Garfinkel AC, Venturini G, Wakimoto H, Repetti G, Alamo L, Sharma A, Agarwal R, Ewoldt JF, Cloonan P, Letendre J, Lun M, Olivotto I, Colan S, Ashley E, Jacoby D, Michels M, Redwood CS, Watkins HC, Day SM, Staples JF, Padrón R, Chopra A, Ho CY, Chen CS, Pereira AC, Seidman JG, Seidman CE. Myosin Sequestration Regulates Sarcomere Function, Cardiomyocyte Energetics, and Metabolism, Informing the Pathogenesis of Hypertrophic Cardiomyopathy. Circulation. 2020;141(10):828-42. Epub 2020/01/28. doi: 10.1161/circulationaha.119.042339. PubMed PMID: 31983222; PMCID: PMC7077965.