By Gaelyn Lyons
At this point, no one is a stranger to COVID-19. We have all been affected by the virus, whether it be socially, physically, or mentally. As of August 15th, there have been about 5.2 million cases in the US, of which about 167,000 patients have died (CDC, 2020). Although it feels as if we’ve been in quarantine for longer than nine months, there is a small bit of hope on the horizon: a coronavirus vaccine.
Over the past couple of months, many researchers around the world have worked tirelessly to develop vaccinations for the virus, and 170 candidates are currently being acknowledged by the World Health Organization (Kommenda et al., 2020). Normally, vaccine development can take anywhere between 10-15 years before being distributed to the public due to intense testing and regulation (The College of Physicians of Philadelphia, n.d.). However, scientists working on a coronavirus vaccine are hoping to have one developed within 12 to 18 months (Kommenda et al., 2020).

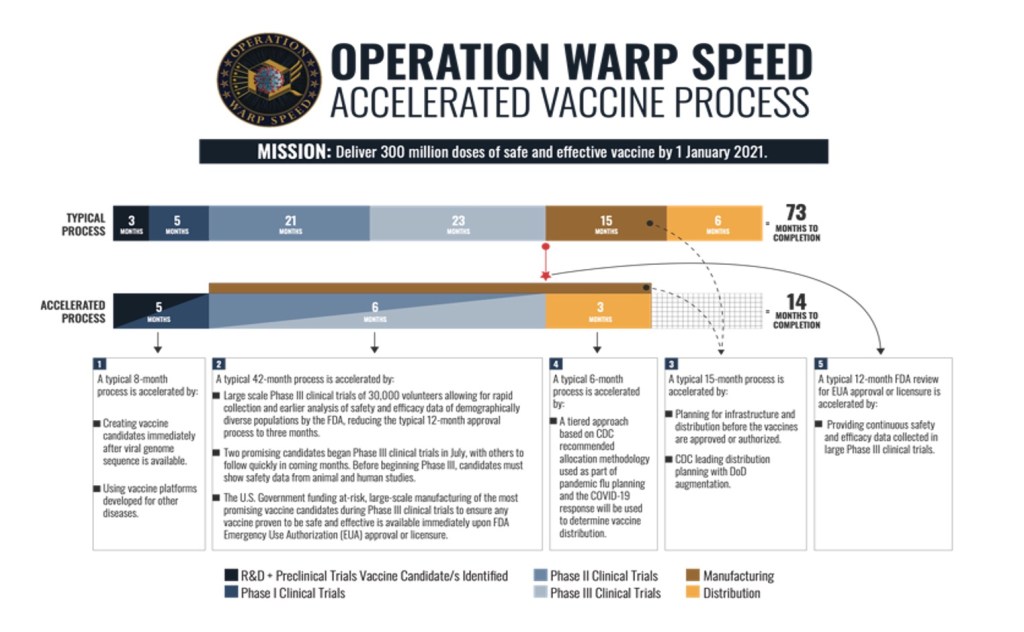
In response to the urgent need of a vaccine, the US government formed Operation Warp Speed (OWS), which is a partnership between public and private companies that is focusing on quickly developing a COVID-19 vaccination that is both safe and effective (U.S. Department of Defense, n.d.). The National Institutes of Health has partnered with 18 pharmaceutical companies and under OWS the US government chose three vaccine candidates to fund for Phase 3 clinical trials: mRNA-1273 from Moderna, AZD1222 from The University of Oxford and Astrazeneca, and BNT162 from Pfizer and BioNTech (Craven, 2020).
Before any new drugs or therapeutics are approved by the FDA, a series of clinical trials must be done to prove the drug is safe and effective (World Health Organization, n.d.). There are four phases in clinical trials. Phase one tests the drug or vaccine in a small group of healthy individuals to determine a safe dose range and any side effects. This is followed by phase two, which tests the drug or vaccine in the population of interest. Phase three is a large-scale trial that tests individuals in different regions and countries to measure consistency of the drug or vaccine. Finally, Phase four takes place when the drug is approved by the FDA, and data is continually collected to measure the long-term effects of the drug or vaccine.
The goal of producing the COVID-19 vaccine is to create herd immunity, where immunizing enough of the population will reduce the spread of the virus (Reynolds, 2020). According to Fauci, in order for the vaccine to be effective, about two-thirds of the population need to be vaccinated, or herd immunity will not be achieved. However, much of the population, including those who have received their mandatory and routine vaccinations, are wary of receiving this future vaccine when it becomes public. Common objections observed on social media include vaccine safety, necessity of a vaccine, trust in the companies producing the vaccine, and uncertainty about the science behind the vaccine (Reynolds, 2020). Misinformation also plays a role in the spread of hesitancy towards the vaccine. Much of the misinformation can be spread via anti-vaxxer groups, wellness communities, and those who may have a political agenda. Choosing to take action against coronavirus has also become a political issue in other countries besides just the US, including France.
Another reason why people question the safety of the COVID-19 vaccine is the short timeline of its development compared to vaccines of the past. In order to speed up the vaccine development process, multiple steps of the clinical phases are combined (Ratanghayra, 2020). This reduces the amount of time it takes to go through each clinical phase while still maintaining the safety and efficacy testing for the COVID-19 vaccine. Recently, Russia came out with a vaccine that was only tested in 76 people and skipped Phase 3 trials (Cohen, 2020 ), (Kommenda et al., 2020). There is some concern of whether the vaccine is truly safe and effective in preventing coronavirus and there is little data provided by Russia to assuage those concerns (Keown, 2020). The US government and pharmaceutical companies working on the COVID-19 vaccine, however, will not skip any of the phases. Instead, these companies will combine the phases together to efficiently and safely develop the vaccine (U.S. Department of Defense, n.d.).
Communication between the government, the scientific community, and the general public is needed now more than ever. The importance of transparency in a time of uncertainty is something that will ultimately benefit people and hopefully lead to the containment of this virus. Overall, the US government and scientists like Dr. Fauci have been helpful in keeping the public informed about where things are when it comes to the development of the COVID-19 vaccine. If anyone is interesting in looking at where clinical trials are for the vaccine or even enrolling in a clinical trial themselves, clinicaltrials.gov is a good resource to use. All this to say, masks need to be worn, social distancing needs to be followed, and an effective and safe vaccine needs to be distributed in order to fight coronavirus. Plus, I just want to eat in a restaurant again.
References
CDC. (2020, August 15). Coronavirus Disease 2019 (COVID-19) in the U.S. Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html
Cohen, J. (2020, August 11). Russia’s approval of a COVID-19 vaccine is less than meets the press release. Science | AAAS. https://www.sciencemag.org/news/2020/08/russia-s-approval-covid-19-vaccine-less-meets-press-release
Craven, J. (2020, August 20). COVID-19 vaccine tracker. https://www.raps.org/news-and-articles/news-articles/2020/3/covid-19-vaccine-tracker
Keown, A. (2020, August 13). Clinical Transparency Proponents Question Safety and Efficacy of Russia’s COVID-19 Vaccine. BioSpace. https://www.biospace.com/article/clinical-transparency-proponents-question-safety-and-efficacy-of-russia-s-covid-19-vaccine/
Kommenda, N., Hulley-Jones, F., Kommenda, N., & Hulley-Jones, F. (2020, August 16). Covid vaccine tracker: When will we have a coronavirus vaccine? The Guardian. https://www.theguardian.com/world/ng-interactive/2020/aug/16/covid-vaccine-tracker-when-will-we-have-a-coronavirus-vaccine
Ratanghayra, N. (2020, July 2). How to Speed Up COVID-19 Vaccine Development. Clinical Lab Manager. https://www.clinicallabmanager.com/trends/infectious-diseases/how-to-speed-up-covid-19-vaccine-development-23156
Reynolds, E. (2020, August 15). Vaccines are safe. But huge numbers of people around the world say they wouldn’t take a Covid jab. CNN. https://www.cnn.com/2020/08/15/health/vaccine-hesitancy-coronavirus-safety-intl/index.html
The College of Physicians of Philadelphia. (n.d.). Vaccine Development, Testing, and Regulation | History of Vaccines. The HIstory of Vaccines. Retrieved August 16, 2020, from /content/articles/vaccine-development-testing-and-regulation
U.S. Department of Defense. (n.d.). Coronavirus: Operation Warp Speed. U.S. Department of Defense. Retrieved August 24, 2020, from https://www.defense.gov/Explore/Spotlight/Coronavirus/Operation-Warp-Speed/
World Health Organization. (n.d.). Clinical trials. Retrieved August 24, 2020, from https://www.who.int/westernpacific/health-topics/clinical-trials
Thank you for your thorough insight and perspective on the Coronavirus pandemic and the updates with current vaccine manufacturing. With the political climate as it is in the U.S., it’s been difficult to get an honest answer from most news stations and talking heads. Keep the communication flowing.
Pingback: The Past, Present, and Future of FDA Accelerate Approval | Lions Talk Science