By Mariam Melkumyan
While certain levels of stress present in our lives is a good thing, sometimes it can be too much. Now is a very stressful time for graduate students; in addition to qualifying and comprehensive exams and dissertations, we are constantly reminded of threats to our health and normal lives due to the COVID-19 pandemic. The inability to continue our experiments, choose a thesis lab, and study without distraction can be very worrying. So why aren’t we all having a breakdown and panic attacks? What is protecting us from this stress and allowing us to function in our daily lives?
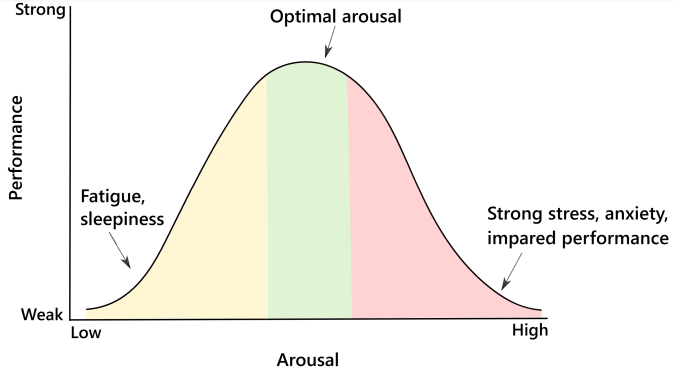
Before delving into the neuroscience of stress, it is important to talk about the Yerkes-Dodson Law. It suggests that there is an optimal level of arousal/stress where we function at our best; too little or too much arousal results in low performance and inability to function.
But why do we need that optimal stress level? Stress allows our bodies to react quickly and be alert. Evolutionarily, it is very important for animals to have some level of stress to run away from predators and hunt for prey. This is done through the fight or flight response and the sympathetic nervous system and hypothalamic-pituitary-adrenal (HPA) axis. When stressed, the hypothalamus sends signals to produce corticotropin releasing factor (CRF) to the pituitary, which then activates the production of adrenocorticotropin hormone (ACTH) leading to the production of cortisol from the adrenal gland. Cortisol then activates our physiological response to stress, such as increased glucose levels, heart rate, and blood flow. However, what protects us from chronic stress is the negative feedback that cortisol sends to its receptors in various parts of the brain, including the hippocampus, hypothalamus, amygdala, and the rest of the body. This stops the cortisol production at its various steps, allowing the body to return to homeostasis. So why doesn’t this protective mechanism work for everyone? Why do some people suffer from chronic stress and anxiety?
Why this mechanism is impaired in anxiety is still largely unknown. Studies point to various genetic variants that might be involved, such as the gene for one of the CRF receptors, preventing it from responding to the negative feedback from CRF2. Other studies suggest single nucleotide polymorphisms in FKBP5, a co-chaperone of the glucocorticoid receptor, resulting in reduced sensitivity to cortisol, which leads to an increased production of the stress hormone and subsequent chronic stress3. In addition to genetic changes, chronic stress has also been shown to increase neuroinflammation, particularly through increased microglia activation leading to elevated pro-inflammatory cytokines4. It is well known that prolonged inflammation can lead to cell damage and death, resulting in multiple disease states.
Since chronic stress can result in not only mental health issues, but also physical health problems, it is important to take time and relax. It is a stressful time and we need to remember to let our body do its job and bring us back to homeostasis.
- Saeed A, Trajanovski S, Van Keulen M, Van Erp J. Deep Physiological Arousal Detection in a Driving Simulator Using Wearable Sensors. In: 2017 IEEE International Conference on Data Mining Workshops (ICDMW). ; 2017:486-493. doi:10.1109/ICDMW.2017.69
- Bradley RG, Binder EB, Epstein MP, et al. Influence of child abuse on adult depression: moderation by the corticotropin-releasing hormone receptor gene. Arch Gen Psychiatry. 2008;65(2):190-200. doi:10.1001/archgenpsychiatry.2007.26
- Binder EB. The role of FKBP5, a co-chaperone of the glucocorticoid receptor in the pathogenesis and therapy of affective and anxiety disorders. Psychoneuroendocrinology. 2009;34 Suppl 1:S186-195. doi:10.1016/j.psyneuen.2009.05.021
- Johnson JD, Campisi J, Sharkey CM, et al. Catecholamines mediate stress-induced increases in peripheral and central inflammatory cytokines. Neuroscience. 2005;135(4):1295-1307. doi:10.1016/j.neuroscience.2005.06.090
This blog has become a valuable resource for me in navigating the world of cognitive enhancement. Thank you for sharing such insightful and practical content!
Khameera Gaozaban Ambri Jawahar
This blog has become an invaluable resource for anyone looking to enhance their cognitive function and support their mental health. Thank you for providing such informative and practical content!
Itrifal Ustukhuddus
Thank you for sharing such valuable insights and empowering readers to prioritize their mental well-being.
Itrifal Ustukhuddus