
Many young scientists imagine themselves curing a disease, dreaming that their thesis work will uncover a disease mechanism and lead to the development of novel therapeutics to help improve patient health. People with loved ones suffering from diseases hope for a cure, too. However, most people do not understand the arduous journey a treatment will take to go from the bench to the patient’s bedside. Each year, a few dozen drugs are licensed by regulatory bodies, such as the U.S. Food and Drug Administration (FDA), but behind those dozen successes are tens of thousands of failed candidate drugs that do not get licensed. Overall, the process of getting a single, safe, and effective drug to market takes approximately 15 years and ranges from $800 million to $3 billion in cost.
The road to discovery typically starts in a basic research laboratory at an academic institution where hard-working graduate students, such as ourselves, investigate the fundamental science of human biology. Once a disease mechanism is discovered, the search for a target molecule to alleviate or cure the symptoms can begin. Over 10,000 compounds are initially evaluated and systematically eliminated through strategic experimentation. This can range from computational modeling to determine how the drug interacts with the target, in vitro tests on cells in culture, and various animal models in vivo. Next, the pre-clinical phase tests about 20 candidate drugs in larger animals in more targeted studies to further identify which drugs would be viable for clinical trials. During this phase, harmful side effects may be discovered, preventing a drug from continuing its journey in the development pipeline. Only half of the drugs tested in the pre-clinical phase will prove to be safe and effective enough to move on to clinical trials.
Clinical trials are the most expensive and time-consuming phase during the course of drug development. About 75% of investor money is spent on clinical trials and FDA approvals. Typically, the first phase of clinical trials tests the safety of small doses of the drug on a group of 20 to 100 healthy volunteers; however, in some cases the process can be expedited and test the drug on the targeted population instead. About half of these drugs (now approximately five drugs) will proceed to Phase 2 clinical trials to be tested in 100 to 500 volunteer patients (people with the disease in question) to determine the most effective dose and delivery method. The majority of drugs that fail in clinical trials fail in Phase 2 because they turn out to be ineffective. A select one or two drugs will advance to Phase 3 trials to be tested on a much larger population of patients. Despite all of the rigorous testing conducted prior to this, about 10% of drugs will still fail at this stage. Once the drug passes Phase 3, the drug can be submitted for FDA approval; however, the journey is not completely over. The FDA often requires that a drug be continually monitored in post-marketing studies (known as Phase IV clinical trials) to ensure its safety for special populations that could not be adequately evaluated during pervious testing, such as pregnant women.
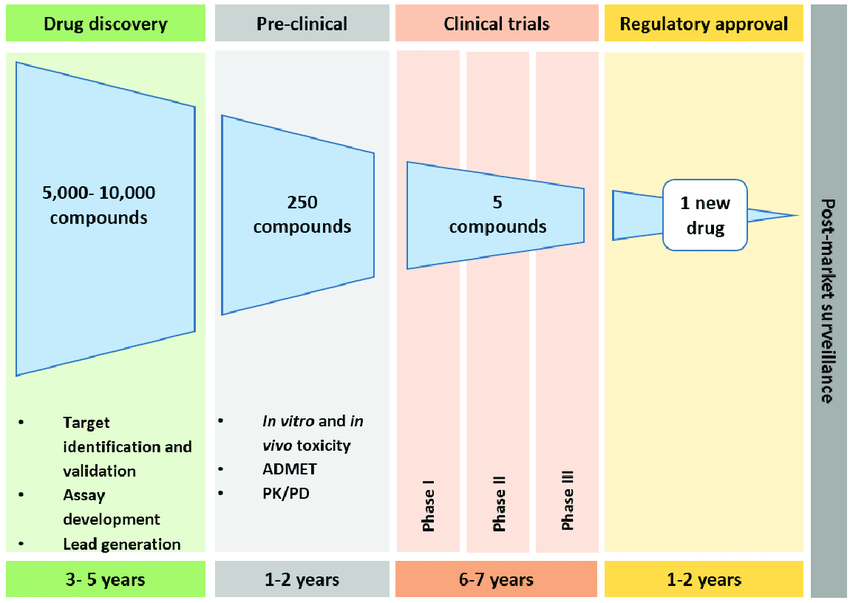
With the rigorous pathway drugs take through development now in mind, it is easier to understand how difficult it is for a drug to make it to market. The diagram above outlines this path for reference. Since January of 2019, the FDA has approved 35 drugs, which is about average for a given year; however, 2020 will be the year to watch. In October, Biogen announced plans to file for licensing of aducanumab, a potential treatment for early Alzheimer’s disease, in early 2020. This announcement has come with much skepticism, which I will address in the new year. Stay tuned!
By Stephanie Baringer
Pingback: The Past, Present, and Future of FDA Accelerate Approval | Lions Talk Science