By: Habiba Abdelhalim
In many animal societies – including humans! – illness can affect more than just the individual that becomes sick. Diseases can spread rapidly amongst members of a group, especially when animals live closely together. Humans regularly implement strategies to mitigate this spread: we stay home when contagious, avoid sick people, and sometimes isolate ourselves until we recover. But in some animal societies, an even more dramatic strategy has evolved: individuals openly signal that they are sick, even when doing so leads to their own death. A recent study1 of ants reveals that infected ant pupae, the developmental stage between larva and adult, can produce chemical signals that effectively tell their nestmates, “I’m infected.” In response, the worker ants destroy the infected individual before the disease can spread through the colony. While this behavior may seem extreme, it demonstrates how self-sacrifice and collective defense shape evolution in social organisms.
Meet the Cast: the garden ant and its remarkable collective defense
The invasive garden ant, Lasius neglectus, lives in large, densely populated colonies in which individuals depend on one another for survival. Workers, sterile females that make up the majority of the colony and carry out nearly all of its day-to-day tasks, unlike the queen whose role is reproduction, gather food, care for young, maintain the nest, and protect the colony from threats. Because so many ants live together in close contact, colonies face a constant risk from pathogens such as bacteria, viruses, and fungi. If a contagious disease were allowed to spread unchecked, it could quickly wipe out the entire colony. To protect themselves, social insects have evolved what scientists call social immunity2. Instead of relying solely on the immune systems of individual ants, the entire colony participates in disease defense through collective behaviors. Worker ants groom one another to remove pathogens, disinfect the nest environment, and sometimes remove infected individuals from the colony. These group-level behaviors function much like an immune system operating at the level of the colony rather than the individual, where worker ants act similarly to immune cells that detect and eliminate threats, and chemical signals between ants resemble signaling molecules like cytokines that coordinate a targeted, collective response to infection.
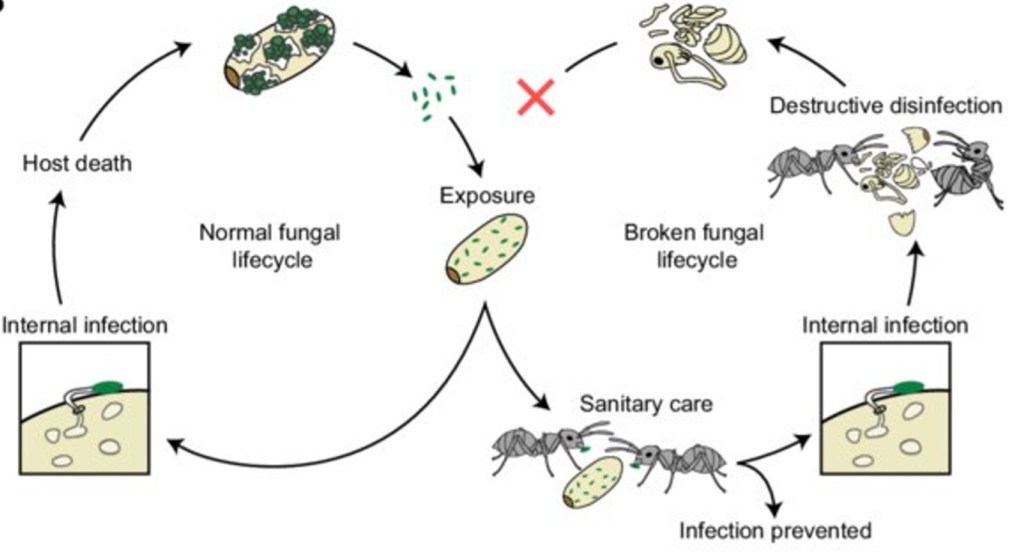
One important stage in an ant’s life cycle, the pupal stage, resembles these behaviors in action. The pupal stage is when developing ants transform from larvae into adults and are usually wrapped inside protective cocoons and cared for by worker ants. Workers frequently move pupae around the nest to maintain the correct temperature and humidity and keep them safe. However, this close care also means that pupae can become a source of infection if they are exposed to pathogens (Figure 1). When a pupa becomes infected, the consequences for the colony can be severe, a single infected individual, handled repeatedly by workers, can rapidly spread disease throughout the nest. This vulnerability makes the pupal stage a critical point in the colony’s disease defense, and a key focus of recent research into how ants detect and respond to infection.
The Big Question: How do ants know when one of their own is sick?
Researchers wanted to understand how ant colonies detect disease and, more importantly, whether infected individuals play an active role in this process1. To appreciate why this question matters, it helps to understand what was previously assumed: for years, scientists assumed that worker ants simply recognized sick pupae through passive changes, such as altered smell from infection. However, the absence of a clear passive signal raised a deeper question that this study set out to answer directly: could infected pupae actively signal their illness to protect the colony? Answering this would reveal whether disease detection in social insects is coordinated, cooperative communication, an insight that could reshape our understanding of collective immunity.
To investigate this, the study examined what happens when ant pupae become infected with the fungal pathogen Metarhizium brunneum. The fungus infects insects by attaching to their outer surface, penetrating their bodies, and eventually killing them7. If allowed to develop fully, it produces spores that can spread to nearby ants, posing a serious threat to the entire colony. Worker ants combat this risk through a behavior known as destructive disinfection3: they remove infected pupae from their cocoons, bite them, and apply antimicrobial substances. Although this kills the infected individual, it prevents the fungus from reproducing and spreading. In this way, sacrificing one ant can protect thousands of others, making early and accurate detection of infection critically important.
However, the researchers1 wondered whether something more active might be happening. Instead of sick ants being passively discovered by workers, could infected pupae actually signal their illness intentionally? If so, the pupae would be participating in the colony’s disease defense by announcing they are sick.
Infected ants send a chemical SOS and their colony listens
To investigate this hypothesis, the researchers infected some pupae with fungal spores while keeping others healthy as controls. They then placed the pupae either alone or together with worker ants and analyzed the chemicals present on the surface of the pupae. Shockingly, the infected pupae changed the chemical composition of their outer surface, producing higher levels of molecules called cuticular hydrocarbons. These compounds form a thin layer on the outside of an ant’s body and are commonly used for communication within colonies. Ants rely on them to recognize nestmates versus intruders, a critical distinction in a densely packed colony where a single misidentification could let a predator or parasite go undetected. Cuticular hydrocarbons also convey social roles, allowing workers to identify whether another ant is a queen, a fellow worker, or a developing pupa in need of care. Beyond identity, cuticular hydrocarbons carry environmental information, helping the colony coordinate responses to changes in temperature, humidity, or threat levels. In short, these molecules are not just a communication tool, they are the foundation of colony organization itself. Even more intriguing was that increased levels of hydrocarbons only occurred when infected pupae were near worker ants. When infected pupae were not near workers, the chemical changes were much less pronounced. The pupae, it seemed, knew they had company, and were tailoring their chemical cry for help accordingly.
Even more remarkable was what happened when researchers took those chemicals and applied them to perfectly healthy pupae. Without any actual infection present, worker ants still sprang into action, removing the pupae from their cocoons and destroying them as if they were a genuine threat. The hydrocarbons alone were enough to trigger the colony’s full defensive response, proving that the chemical signal, not the infection itself, is what drives the colony to act.
A genetic answer to a behavioral question
This discovery raises an important question: why would an organism deliberately produce a signal that leads to its own destruction?
The answer, as it turns out, lies in the evolutionary structure of ant colonies.
Most ants in a colony are closely related, sharing many of the same genes. Worker ants usually do not reproduce themselves; instead, they help raise the offspring of the queen. Because of this close genetic relationship, helping relatives survive can indirectly pass on an individual’s genes. Biologists describe this concept as inclusive fitness4, where natural selection favors behaviors that increase the survival and reproduction of genetically related individuals, even if those behaviors harm the individual performing them. In the case of infected pupae, signaling their illness allows the colony to eliminate a dangerous infection early. Although the signaling pupa dies, the action protects many relatives that share its genes. This biological principle extends well beyond the ant kingdom into animals, with meerkats5 taking turns acting as sentinels, watching for predators and sounding an alarm to warn the group, even though doing so increases their own risk.
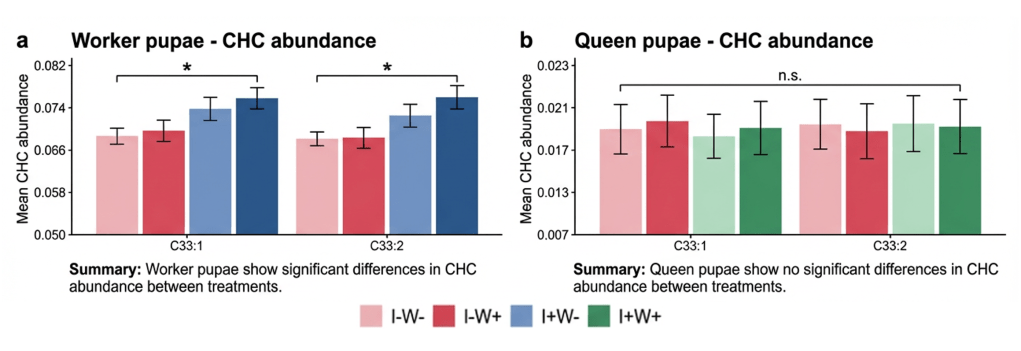
Interestingly, the researchers discovered that not all pupae produce this cuticular hydrocarbon signal. Ant colonies produce two types of female individuals: workers, who make up the vast majority of the colony and perform all day-to-day tasks, and future queens, who are reared specifically to leave the colony, mate, and found entirely new colonies of their own. While workers are essentially replaceable, a queen represents the colony’s entire reproductive future, her survival carries far greater long-term consequences for the colony’s success. Worker pupae displayed the illness signal when infected, but queen pupae did not. At first glance, this difference might appear selfish – perhaps future queens were hiding their illness, to avoid being killed! However, additional experiments provided a different explanation.
The scientists measured the strength of the immune response in both types of pupae and tracked how the infection developed over time. Interestingly, they found that queen pupae had stronger immune defenses and were better able to control the fungal infection. While the infection continued to grow in worker pupae, queen pupae often reduced their pathogen levels after an initial increase (Figure 2). In other words: queen pupae had a realistic chance of recovering from the infection on their own. Because queen pupae were more likely to survive, it would be wasteful for them to signal for destruction too early and, by prematurely terminating, potentially depriving the future colony whatever collective benefits may be achieved from their healthy birth.
Key findings
The cuticular hydrocarbon chemical signaling system therefore appears to be finely tuned. Pupae signal their illness primarily when the infection becomes overwhelming and their immune system can no longer stop the pathogen. This ensures that the colony eliminates individuals that are unlikely to recover, while allowing others to survive if possible.
The chemical signals themselves, furthermore, are surprisingly precise: they involve specific hydrocarbons that only differ slightly in their molecular structure. Worker ants can detect these subtle chemical differences and respond accordingly. The sensitivity highlights the sophistication of chemical communication in ant societies, where tiny variations in scent can convey complex information.
Similar behaviors have been observed in other social insects. In honeybee colonies, for example, worker bees remove larvae that show signs of disease, preventing infections from spreading through the hive6. Although the exact chemical signals differ between species, the general principle is the same: social insects rely on chemical communication to identify and remove infected individuals before pathogens can spread.
Zooming out: connecting ant behavior to patterns in human bodies and society
Social immunity behavior also resembles processes that occur within the bodies of multicellular organisms. In humans and other animals, damaged or infected cells sometimes signal immune cells to destroy them, preventing infections or cancer from spreading through the body. In a similar way, an ant colony can be thought of as a superorganism, where individual ants function like cells within a larger body. Just as immune cells remove damaged cells in a body, worker ants remove infected individuals in the colony. Ultimately, Dawson et al.’s1 research reveals a powerful lesson about cooperation in nature. Even the smallest creatures can demonstrate complex forms of communication and self-sacrifice. By signaling when they are sick, these ants help maintain the health of their entire society. What might appear to be a tragic end for one individual becomes a triumph for the colony as a whole.
TL; DR:
- Infected ant pupae release chemical signals prompting nestmates to destroy them, preventing colony-wide spread.
- Future queens are not destroyed, relying on stronger immunity instead.
- Social immunity behaviors reveal ant colonies functioning as a superorganism, with individuals sacrificing themselves for collective survival.
Reference
- Dawson, E. H., Hoenigsberger, M., Kampleitner, N., Grasse, A. V., Lindorfer, L., Robb, J., Beikzadeh, F., Strahodinsky, F., Leitner, H., Rajendran, H., Schmitt, T., & Cremer, S. (2025). Altruistic disease signalling in ant colonies. Nature communications, 16(1), 10511.
- Malagocka, J., Eilenberg, J., & Jensen, A. B. (2019). Social immunity behaviour among ants infected by specialist and generalist fungi. Current opinion in insect science, 33, 99–104.
- Pull, C. D., Ugelvig, L. V., Wiesenhofer, F., Grasse, A. V., Tragust, S., Schmitt, T., Brown, M. J., & Cremer, S. (2018). Destructive disinfection of infected brood prevents systemic disease spread in ant colonies. eLife, 7, e32073.
- Hamilton W. D. (1964). The genetical evolution of social behaviour. I. Journal of theoretical biology, 7(1), 1–16.
- Young, A. J., Spong, G., & Clutton-Brock, T. (2007). Subordinate male meerkats prospect for extra-group paternity: alternative reproductive tactics in a cooperative mammal. Proceedings. Biological sciences, 274(1618), 1603–1609.
- Wagoner, K. M., Millar, J. G., Schal, C., & Rueppell, O. (2020). Cuticular pheromones stimulate hygienic behavior in the honey bee (Apis mellifera). Scientific reports, 10(1), 7132.
- Csata, E., Pérez-Escudero, A., Laury, E., Leitner, H., Latil, G., Heinze, J., Simpson, S. J., Cremer, S., & Dussutour, A. (2024). Fungal infection alters collective nutritional intake of ant colonies. Current biology : CB, 34(4), 902–909.e6.