By Ryan Hylton, PhD
This article is based on the dissertation of Dr. Ryan Hylton who defended his thesis at PSCOM on May 16, 2022.
The advantages of cryo-electron tomography
In cell biology publications, many mechanistic models are built on a research team’s interpretations of relatively indirect observations. For instance, changes in protein expression levels are determined without direct visualization of the protein of interest. Fluorescent microscopy can reveal the rough morphology of intracellular compartments and the localization of proteins, but in an indirect manner through multiple antibodies. Even in structural biology, individual proteins are directly visualized, but are generally purified and observed outside of a cell. The structure of these proteins can be determined with high precision through various methods, but there is always a question as to whether the protein’s structure or mechanism of action behaves similarly in the cell.
Cellular machinery can instead be directly visualized in its native environment through the use of cryo-electron tomography (cryo-ET). Here, cells are grown on electron microscopy (EM) grids before being plunge-frozen and imaged in a transmission electron microscope. The resulting images facilitate analysis of mesoscale cytoplasmic architecture and nanoscale structure determination.
The fact that the cell’s interior can be visualized directly with high resolution makes cryo-ET an ideal technique for exploring cell types and subcellular compartments that are poorly understood. This strategy has been employed by other labs, particularly those studying the microbial world where there are so many organisms, many of which have undiscovered ultrastructure that lies within them. The work that comprised my dissertation had a similar theme and is a good example of the scientific advancements that come from simply exploring the unknown.
The growth cone
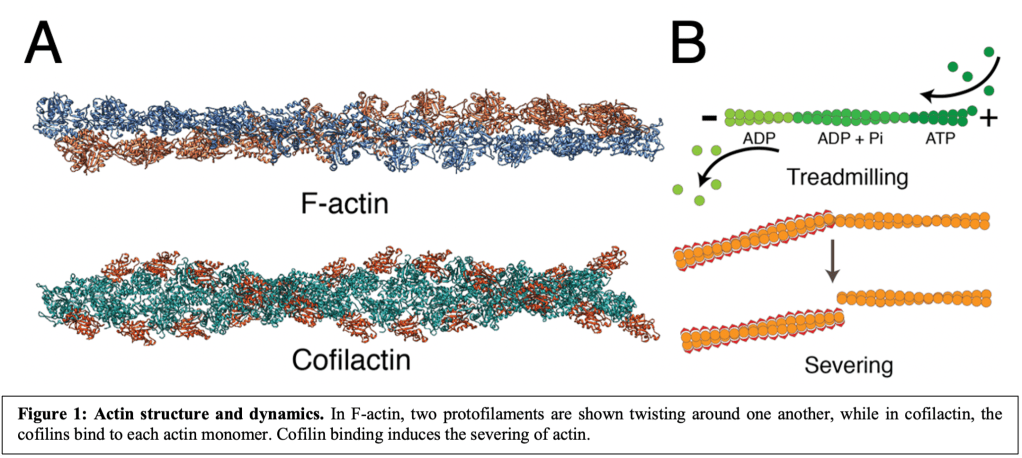
I did my PhD work in Dr. Matthew Swulius’ lab at the Penn State College of Medicine where we use cryo-ET to study neurodevelopment. Mature neurons communicate through synapses that lie on projections called neurites. These neurites find their eventual synaptic partners during neurodevelopment through the action of a subcellular compartment at their distal tip called the growth cone1,2. The growth cone is a hand-shaped structure with finger-like projections called filopodia that act as antennae, finding and responding to extracellular cues1,2. Filopodia are largely comprised of the helical polymer filamentous actin (F-actin)1,3 (Fig. 1A).
Therefore, filopodial size, shape, and motility are determined by actin dynamics (Fig. 1B), which themselves are regulated, in large part, by a series of actin binding proteins4. For instance, the primary focus of my dissertation was the actin binding protein cofilin. Cofilin binds to actin in need of recycling where it induces filament severing5-8 (Fig. 1A). This severing occurs at the boundary between cofilin-decorated actin (often referred to as “cofilactin”) and bare actin filament segments7(Fig. 1B).
Growth cones have been studied by light microscopy for decades and much is known about the extracellular ligands and intracellular signaling cascades that control their movements1,2. Comparatively little is known about their mesoscale ultrastructure. They have been observed by EM9-11, but almost exclusively using traditional methods that involve plastic embedding and freeze substitution, methods of preservation that tend to subtly alter the localization and structure of proteins and membrane compartments. So, we sought to elucidate the ultrastructure of the growth cone cytoskeleton using cryo-ET, which preserves the cytoplasm and its constituents in a near-native, frozen-hydrated state. Importantly, when we began this investigation, we didn’t know what we would find. Although we didn’t start with a specific hypothesis worth testing, what we discovered acted as a seed that germinated into a multifaceted research program with many arms, each of which now have hypotheses that the Swulius lab intends to test in the coming years.
A hyper-twisted filament
When I examined growth cones (Fig. 2A), I found them to be filled with F-actin, consistent with previous literature. In the body of the growth cone were many individual actin filaments in a branched network, while actin in the filopodial tips was organized into tightly packed bundles (Fig. 2B). However, near the proximal base of the filopodia I found bundles of actin filaments that looked different than normal actin (Fig. 2C). This new filament was wider than actin, but most strikingly, its helical crossover distance was smaller compared to actin. Therefore, we described this new filament as “hyper-twisted”.
Bundles of cofilactin in growth cone filopodia
A search of the literature revealed that actin filaments fully decorated in cofilin take up this hyper-twisted conformation12,13. Importantly, this had only been shown in vitro, with purified actin and cofilin; meaning this would be the first in situ visualization of this structure. For confirmation, I turned to immunofluorescence where I expected heavy cofilin localization near the base of growth cone filopodia. After significant troubleshooting, which revealed differential staining with different permeabilization reagents, I confirmed my hypothesis. To stain filopodial tips, I labeled fascin, a protein that crosslinks F-actin in filopodia into tightly-packed bundles of linear filaments14,15. To our surprise, the resulting images showed a clear inverse correlation between fascin and cofilin along the length of filopodia, with fascin almost exclusively being found in the distal tips and cofilin in the proximal bases. This indicated to us that fascin may not play a significant role in the crosslinking of cofilactin bundles in growth cone filopodia, which was further supported by our tomography data.
Cofilactin bundles facilitate lateral space searching in a subset of filopodia
To next determine the function of the cofilactin bundles in filopodia, I turned to live-cell imaging and obtained movies with a particular emphasis on filopodial dynamics. I found that filopodial movements tended to hinge upon the transition region between the cofilactin and normal actin bundles (Fig. 3).
I then quantified the lateral movements of each filopodial region (actin, transition, and cofilactin) and found that two general phenotypes existed: “searching” and “resting”. In searching filopodia, the actin-rich tip moved most, and they appeared as though they were actively searching their environment for cues (Fig. 3B & D). In resting filopodia, the tips were relatively stable, and most movement came from the cofilactin-rich base (Fig. 3A & C). In both cases, however, the transition region moved minimally, suggesting this region acts as a hinge about which the more distal and proximal filopodial regions rotate. Our hypothesis as to why this is the case is that the filopodial tips are crosslinked by fascin, and therefore are relatively inflexible. Similarly, cofilactin filaments in the filopodial bases clearly appear to be crosslinked due to their high degree of organization and tight filament packing, but through a currently unknown mechanism. In the transition region, however, cofilin binding works to exclude fascin, but cofilactin filaments are too sparse to significantly link together. This results in a region with a high degree of flexibility due to the lack of crosslinkers, which enables it to act as a hinge point during filopodial space searching (Fig. 4).
Conclusions
There are, of course, many open questions that remain. However, this work provides evidence for a new role for cofilin beyond simply severing actin filaments. This discovery was made possible by at least two factors: 1) the mere act of exploring growth cones in a way they had not been observed previously and 2) the ability to visualize growth cone ultrastructure in a largely unperturbed state using cryo-ET. Fortunately, we were able uncover something that we did not expect and this has led to many hypotheses worth testing in a more systematic fashion. In so doing, the Swulius lab plans to determine the mechanism of cofilactin crosslinking in filopodia, how cofilactin bundles are regulated, the precise mechanism by which the filopodial transition region acts as a hinge point during space searching, among other things. Ideally, this line of research will aid future understanding of neurodevelopmental disorders and how we might treat them, as well as how neuroregeneration works and how it can be manipulated/enhanced to improve recovery from injury.
*Figures are borrowed from my written dissertation entitled: “Cofilactin bundling in neurodevelopment and degeneration”.
**More information can be found from our recent article with the following citation:
Hylton, R.K., Heebner, J.E., Grillo, M.A. et al. Cofilactin filaments regulate filopodial structure and dynamics in neuronal growth cones. Nat Commun 13, 2439 (2022). https://doi.org/10.1038/s41467-022-30116-x
TL:DR
- Neurites navigate their environment through the action of the neuronal growth cone, a subcellular compartment whose ultrastructure is poorly understood.
- Cofilin is an actin binding protein that plays a role in growth cone motility by severing and recycling actin filaments.
- I discovered that cofilin-decorated actin filaments and normal actin filaments occupy distinct segments of growth cone filopodia and space between these areas acts as a hinge that enables lateral movement during space searching.
References
- Lowery, L. A. & Vactor, D. V. The trip of the tip: understanding the growth cone machinery. Nat Rev Mol Cell Bio 10, 332–343 (2009).
- Vitriol, E. A. & Zheng, J. Q. Growth Cone Travel in Space and Time: The Cellular Ensemble of Cytoskeleton, Adhesion, and Membrane. Neuron 73, 1068–1081 (2012).
- Dent, E. W., Gupton, S. L. & Gertler, F. B. The Growth Cone Cytoskeleton in Axon Outgrowth and Guidance. Csh Perspect Biol 3, a001800 (2011).
- Pollard, T. D. Actin and Actin-Binding Proteins. Csh Perspect Biol 8, a018226 (2016).
- Carlier, M.-F. et al. Actin Depolymerizing Factor (ADF/Cofilin) Enhances the Rate of Filament Turnover: Implication in Actin-based Motility. J Cell Biology 136, 1307–1322 (1997).
- Maciver, S. K., Zot, H. G. & Pollard, T. D. Characterization of actin filament severing by actophorin from Acanthamoeba castellanii. J Cell Biology 115, 1611–1620 (1991).
- Suarez, C. et al. Cofilin Tunes the Nucleotide State of Actin Filaments and Severs at Bare and Decorated Segment Boundaries. Curr Biol 21, 862–868 (2011).
- Andrianantoandro, E. & Pollard, T. D. Mechanism of Actin Filament Turnover by Severing and Nucleation at Different Concentrations of ADF/Cofilin. Mol Cell 24, 13–23 (2006).
- Yamada, K. M., Spooner, B. S. & Wessells, N. K. Ultrastructure and function of growth cones and axons of cultured nerve cells. J Cell Biology 49, 614–635 (1971).
- Gordon-Weeks, P. R. The ultrastructure of the neuronal growth cone: New insights from subcellular fractionation and rapid freezing studies. Electron Microsc Rev 1, 201–219 (1988).
- Tsui, H. C., Ris, H. & Klein, W. L. Ultrastructural networks in growth cones and neurites of cultured central nervous system neurons. Proc National Acad Sci 80, 5779–5783 (1983).
- McGough, A., Pope, B., Chiu, W. & Weeds, A. Cofilin Changes the Twist of F-Actin: Implications for Actin Filament Dynamics and Cellular Function. J Cell Biology 138, 771–781 (1997).
- Tanaka, K. et al. Structural basis for cofilin binding and actin filament disassembly. Nat Commun 9, 1860 (2018).
- Yang, S. et al. Molecular mechanism of fascin function in filopodial formation. J Biological Chem 288, 274–84 (2012).
- Vignjevic, D. et al. Role of fascin in filopodial protrusion. J Cell Biology 174, 863–875 (2006).
- Riedl, J. et al. Lifeact: a versatile marker to visualize F-actin. Nat Methods 5, 605–607 (2008).