By Wenxue Lin, PhD
This article is based on the dissertation of Dr. Wenxue Lin who defended his thesis at PSCOM on April 27, 2022.
Although the prevalence of cigarette smoking has decreased from 20.9% (2005) to 14.0% (2019),1,2 cigarette smoking still remains the primary preventable cause of death in the United States and is responsible for more than 480,000 deaths annually.3 In 2018, the Food and Drug Administration declared that reducing nicotine in tobacco products in an effort to make them minimally addictive or nonaddictive was a priority.4 Very low nicotine content (VLNC) cigarettes (0.2 mg nicotine/ cigarette or 0.4 mg of nicotine per gram of tobacco) are considered as nonaddictive.5,6 I wanted to test whether there were any differences in reduced nicotine content compared to usual nicotine content in terms of smoking satisfaction as measured by the modified Cigarette Evaluation Questionnaire (mCEQ).7
What did I do?
A double-blind, parallel group randomized clinical trial was conducted at the Penn State College of Medicine (Hershey, Pennsylvania) and George Washington University (Washington, D.C.) between 2015-2018. The study consisted of four phases (Baseline 1, Baseline 2, Randomized, and Treatment Choice) with 11 clinical visits at the study centers over 33 weeks (Figure 1).5,6
In the 18-week Randomized phase, participants were randomized to the control arm in which they used the Usual Nicotine Content (UNC) study cigarettes, or the intervention arm to receive progressively Reduced Nicotine Content (RNC) study cigarettes.5 Participants in the control group received research cigarettes with 11.6 mg nicotine/cigarette for the entire 18-week period. Participants in the gradual group received research cigarettes with nicotine contents 7.4 (visit 4), 3.3 (visit 5), 1.4(visit 6), 0.7 (visit 7) and 0.2 (visit 8 and visit 9) mg nicotine/cigarette.5,6 During the randomization phase, participants in both UNC and RNC arms completed the modified Cigarette Evaluation Questionnaire (mCEQ),7 with a focus on smoking satisfaction score, to compare the acceptability and satisfaction of study cigarettes between UNC and RNC groups.
Figure 2 shows the comparison of mean scores between RNC and UNC on mCEQ subscale 1 from visit 4 to visit 9. Table 1 lists mCEQ subscale 1 (Satisfaction) mean differences between RNC and UNC group with p-value. For Satisfaction subscale, the rating score for RNC was higher than UNC at visit 4 and 5 (RNC nicotine content: 7.4 mg and 3.3 mg, respectively), but starting from visit 6 (nicotine content: 1.4mg), RNC shows significantly lower rating score in satisfaction compared to UNC (Table 1). Overall, RNC group was equally satisfied until visit 6, which correlated with an almost 10-fold reduction of the nicotine content in their cigarettes.
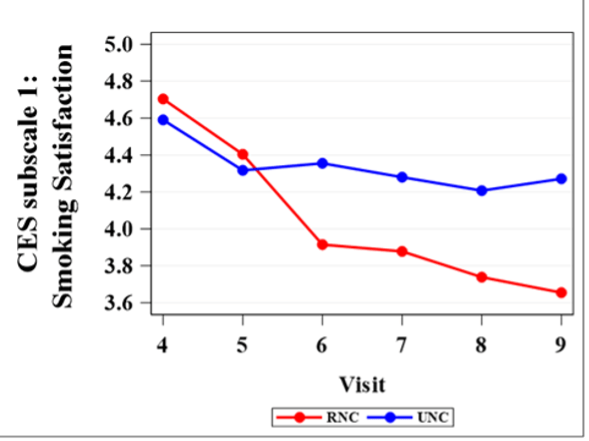
Table 1. Results of analyses comparing mCEQ subscales in the RNC vs. UNC groups
| No. of Visit | Visit 4 | Visit 5 | Visit 6 | Visit 7 | Visit 8 | Visit 9 |
| Nicotine Content | 7.4 mg | 3.3 mg | 1.4 mg | 0.7 mg | 0.2 mg | 0.2 mg |
| p-value | p-value | p-value | p-value | p-value | p-value | |
| Satisfaction1 | 0.5214 | 0.6304 | 0.0172 | 0.0356 | 0.0181 | 0.0026 |
Why is this important?
Smoking Satisfaction is an important way to reflect smokers’ acceptability of reduced nicotine products. I found that RNC smokers rated significantly lower scores in smoking satisfaction. Visit 6 is usually the start point of where there were significant differences in smoking satisfaction between two treatments. The nicotine content for this visit is 1.4 mg nicotine per cigarette.9 Thus, visit 6 or 1.4 mg nicotine content may play a vital role in participants’ rating in satisfaction scale. It is important for researchers to come up with a possible solution to make a smooth transition from 1.4 mg nicotine content cigarette to Very Low Nicotine Content (VLNC; nicotine content: 0.2 mg/cigarette) with better compliance and lower rates of participant attrition. The findings from our study revealed that 1.4 mg nicotine content can be the key point for future Very Low Nicotine Content cigarettes transition research.9
A lower satisfaction score is usually considered as a double-edged sword. On the one hand, smokers who are not satisfied with reduced nicotine research cigarettes may quit smoking cigarettes, but on the other, addicted smokers may have increased need for normal nicotine content cigarettes or switch to other high nicotine content tobacco products.8 Understating the impact of nicotine reduction on smoking satisfaction through cigarette evaluation would provide valuable information and possible direction to the FDA on implementing a nicotine reduction policy for cigarettes.9
If you are interested in reading a more in-depth and scientific explanation of RNC subjective cigarette ratings research, this is published and publicly available at:
Lin, Wenxue, Nicolle M. Krebs, Junjia Zhu, Jonathan Foulds, Kimberly Horn, and Joshua E. Muscat. 2020. “Comparison between Gradual Reduced Nicotine Content and Usual Nicotine Content Groups on Subjective Cigarette Ratings in a Randomized Double-Blind Trial” International Journal of Environmental Research and Public Health 17, no. 19: 7047. https://doi.org/10.3390/ijerph17197047
If you or a loved one suffers from addiction please call The National Drug Helpline at (844)289-0879 for a free and private call to get information on substance use or misuse.
TL:DR
The impact of reducing nicotine consumption through smoking satisfaction is important due to the remaining prevalence of cigarette smoking. I compared the participants’ smoking satisfaction between gradually reducing nicotine content and usual nicotine content. I found a critical point in which the participants reducing their nicotine content became unsatisfied, indicating a need for further intervention.
References
1. Cornelius ME, Wang TW, Jamal A, Loretan CG, Neff LJ. Tobacco Product Use Among Adults—United States, 2019. Morbidity and Mortality Weekly Report. 2020;69(46):1736.
2. Jamal A, Phillips E, Gentzke AS, et al. Current cigarette smoking among adults—United States, 2016. Morbidity and Mortality Weekly Report. 2018;67(2):53.
3. US Department of Health and Human Services. “The health consequences of smoking—
50 years of progress: a report of the Surgeon General.” 2014.
4. Food and Drug Administration. Tobacco Product Standard for Nicotine Level of Combusted Cigarettes: 83 FR 11818; Federal Register: Washington, DC, USA, 2018; pp. 11818–11843.
5. Krebs NM, Allen SI, Veldheer S, et al. Reduced nicotine content cigarettes in smokers of low socioeconomic status: study protocol for a randomized control trial. Trials. 2017;18(1):1-13.
7. Cappelleri JC, Bushmakin AG, Baker CL, Merikle E, Olufade AO, Gilbert DG. Confirmatory factor analyses and reliability of the modified cigarette evaluation questionnaire. Addictive behaviors. 2007;32(5):912-923.
8. Smith TT, Donny EC, Luo X, et al. The impact of gradual and immediate nicotine reduction on subjective cigarette ratings. Nicotine and Tobacco Research. 2019;21(Supplement_1):S73-S80.
9. Lin W, Krebs NM, Zhu J, Foulds J, Horn K, Muscat JE. Comparison between Gradual Reduced Nicotine Content and Usual Nicotine Content Groups on Subjective Cigarette Ratings in a Randomized Double-Blind Trial. International Journal of Environmental Research and Public Health. 2020;17(19):7047.
