By Cole Burgess
Multiple sclerosis (MS) is an auto-immune disease of the central nervous system (CNS) that involves auto-reactive lymphocytes (simplified to B cells). MS affects 400,000 people in the United States and nearly 2 million people worldwide, according to a study published in 20181. MS occurs more commonly in women than men at a rate of 1.5 to 2.5:12. Throughout the progression of MS are multiple inflammatory events in which the myelin sheath coating neurons deteriorate, leading to the development of brain lesions, or patches of damaged brain tissue. In a healthy individual, B cells produce antibodies that bind pathogens and toxic products throughout the body. During antibody production, B cells undergo somatic hypermutation to generate new antibodies that can bind to target proteins with higher affinity. More specifically, somatic hypermutation is the rearrangement of gene fragments to create a new gene sequence, unique from the original germline sequence. B cells will continue through this process to produce antibodies with increased specificity to the target protein. In the case of MS, these B cells have a defect that leads to the production of self-targeting antibodies, causing the patient to mount an immune response to its own products. This self-response, or auto-immunity, during MS leads to high levels of inflammation in the CNS and perpetuates the loss of myelin sheaths. There are currently no known causes of MS, although, numerous risk factors have been suggested, such as smoking, obesity, and low vitamin D levels3. Additionally, several viruses have been implicated in the development of MS, such as mumps, measles, varicella-zoster virus, and Epstein-Barr virus (EBV)4. Recent work has shed more light on the extent of EBV involvement in MS development through molecular mimicry, the production of viral proteins similar to the host.
To understand how molecular mimicry can occur between EBV proteins and host proteins in MS, it is important to know how EBV infection progresses. EBV is a ubiquitous pathogen, with some estimates that nearly 90% of all adults are infected by the virus2. Most individuals are infected with EBV at a young age, during adolescence, or less commonly during adulthood. The virus is typically passed through the saliva of infected individuals and is transmitted to a new host where it can establish an infection within B cells (Fig. 1)5. Infected cells are detected by the immune system and are killed to prevent further infection. In response to the immune system, EBV will silence its genes and enter a dormant stage, also known as restricted latency, where only two gene products are produced (EBNA1 and EBER RNAs). The function of the EBER RNAs remains unsolved despite high levels of expression in infected cells. EBNA1 allows for the virus to replicate during B cell division to ensure that the virus propagates along with its infected cell. Infected individuals develop an anti-EBNA1 antibody response that targets infected cells5. In summary, EBV enters a host through the saliva and infects B cells, and within the infected B cells, EBV enters a dormant stage where EBNA1 is expressed to maintain the virus during host cell proliferation.
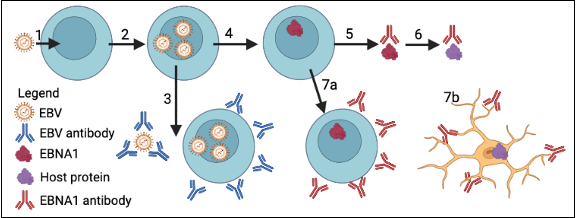
In a recent study, EBNA1 was proposed to be a factor in the development of MS through molecular mimicry4. During molecular mimicry, autoimmunity is established when an immune response targeting a foreign antigen cross-reacts with a host protein that shares a similar structure6. Lanz et al. propose EBNA1-mediated autoimmunity occurs via structural components shared by EBNA1 and the host protein GlialCAM4. GlialCAM is a protein that facilitates cell adhesion and intracellular trafficking for glial cells located in the CNS and epithelial cells in the liver7. In the study, an antibody from a patient was isolated (MS39p2w174) that binds to EBNA1 and GlialCAM. Through structural analysis, it was determined that EBNA1 and GlialCAM share a central epitope (binding region) for MS39p2w174 (the autoreactive antibody). Crucially, the study showed that germline antibodies from the same MS patient had significantly less affinity to GlialCAM than MS39p2w174. Therefore, in the MS patient, somatic hypermutation of their B cells led to the MS39p2w174 autoreactive antibody that strongly interacted with the host protein GlialCAM. The autoreactive antibody can lead to an immune response to GlialCAM-expressing-cells in the CNS, leading to inflammation and deterioration of myelin sheaths. These findings provide a concrete example of molecular mimicry that is detectable in MS patients but not in healthy individuals. Their results from this MS patient were consistent with others in their study cohort, where 7 out of 9 MS patients had antibodies that were autoreactive to GlialCAM. Additionally, in a larger cohort with 71 MS patients and 50 healthy controls, the trend remained constant4.
Within the fields of virology and immunology, this paper has caused a significant amount of controversy. Prior to the Lanz et al. publication, there was only evidence that EBV provided an indirect mechanism to MS development, similar to other described risk factors3. Previous studies have shown that EBV infection during adolescence increases the risk of developing MS by 2 to 3-fold. In addition, EBV infection has been shown to occur before MS onset in multiple studies2. It is possible that EBV infection can lead to the indirect activation of autoimmunity, but Lanz et al. provide evidence of a direct link between EBV and autoimmunity in MS patients through molecular mimicry. The development of autoreactive antibodies that target GlialCAM after EBV infection, may lead to autoimmune-inflicted damage in the CNS of MS patients. These findings beg the question “if EBV infects the majority of the population, why do only certain individuals develop MS?” Some theories have arisen that connect EBV with other factors such as HLA gene type, obesity, and timing of infection, among others3. Unfortunately, there is no obvious answer to this question, and future research is required to explore the role of EBV in MS development. MS remains a complex disease with unresolved origins, although EBV molecular mimicry may provide insight into disease progression.
TL:DR
EBV molecular mimicry through EBNA1 protein leads to autoimmunity targeting GlialCAM in glial cells of the CNS, providing a connection between EBV and MS onset.
References
1. Cotsapas, C., M. Mitrovic, and D. Hafler, Multiple sclerosis. Handb Clin Neurol, 2018. 148: p. 723-730.
2. Ascherio, A. and K.L. Munger, Epidemiology of Multiple Sclerosis: From Risk Factors to Prevention-An Update. Semin Neurol, 2016. 36(2): p. 103-14.
3. Olsson, T., L.F. Barcellos, and L. Alfredsson, Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis. Nat Rev Neurol, 2017. 13(1): p. 25-36.
4. Lanz, T.V., et al., Clonally expanded B cells in multiple sclerosis bind EBV EBNA1 and GlialCAM. Nature, 2022. 603(7900): p. 321-327.
5. Longnecker, R., E. Kieff, and J. Cohen, Fields Virology. 2013: p. 1898-1954.
6. Cusick, M.F., J.E. Libbey, and R.S. Fujinami, Molecular mimicry as a mechanism of autoimmune disease. Clin Rev Allergy Immunol, 2012. 42(1): p. 102-11.
7. Barrallo-Gimeno, A. and R. Estevez, GlialCAM, a glial cell adhesion molecule implicated in neurological disease. Adv Neurobiol, 2014. 8: p. 47-59.