By Xingran Weng, DrPH
This article is based on the dissertation of Dr. Xingran Weng who defended his thesis at PSCOM on February 22, 2022.
Let’s think about a tough situation
Imagine an older individual has been diagnosed with cancer and moderate to severe Alzheimer’s disease (AD) (roughly 20% older persons with cancer diagnosed with dementia).1,2 The cancer prognosis is not optimal. Also, this individual has not yet discussed any treatment plans/goals with family members while the individual continuously exhibits various psychological and behavioral symptoms at home. Family members are frustrated and cannot properly handle this individual’s symptoms due to AD and cancer. This individual has been brought to the hospital more frequently lately. The medical team starts to encounter great difficulties when evaluating this person’s overall condition since AD has significantly compromised this individual’s cognition and communication skill. Consequently, appropriately planning for this person’s End-of-Life (EOL) care becomes critical yet extremely challenging for healthcare providers and family members.
Why End-of-Life care?
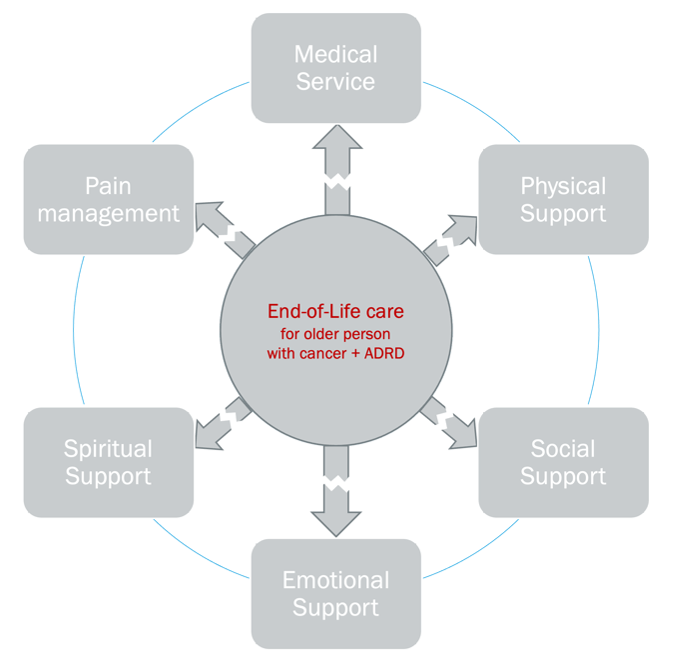
EOL care consists of medical, physical, social, emotional, and spiritual support that can be provided to individuals with terminal cancer to reduce symptoms and pain.3,4 When a person’s medical team deems the cancer is no longer manageable, additional curative and invasive treatment may be replaced by comfort-based supports based on the patient’s goals and preferences in order to avert excessive discomfort.5,6 Nevertheless, EOL care goals sometimes will be sub-optimal if aggressive healthcare services are requested (i.e., using ventilation/intubation or ER/ICU to prolong life expectancy, receiving invasive procedures/more potent medications to potentially cure cancer, and utilizing aggressive resuscitation).7 Receiving aggressive EOL care can significantly impair patients’ quality of life.8,9 Older persons with cancer are especially in a vulnerable position at EOL where their care preferences are often neglected because they are usually deemed less competent to make medical decisions due to their age and disease status.10 Also, the presence of other chronic conditions could complex those older individuals’ treatment outcome at the EOL.11
Alzheimer’s Disease and Related Dementias (ADRD) progressively and irreversibly compromises an individual’s memory, cognitive abilities, and physical functions with no effective treatment to cease its progression.12-14 As ADRD progresses, older persons with both cancer and ADRD start to face various challenges (less responsive to treatment and compromised communication skills) while planning for their EOL care (Figure 1).12,15-19 Eventually, the quality of their EOL care may be severely compromised.
Since EOL care is a critical component throughout the care continuum and associated with patients’ quality of life, I initially thought there should be plenty of scientific evidence of assessing the EOL care quality among individuals with both cancer and ADRD. However, scarce studies had focused on assessing the quality or patterns of EOL among that particular group. To further reveal the lack of attention of EOL care in the field of cancer care, the National Cancer Institute funded nearly $250 million (FY 2018) for studies on cancer screening, prevention, treatment, and survivorship, which was 19 times the spending on studies investigating the EOL care among persons with cancer.20 Although the Institute of Medicine has been advocating for the advancement of EOL care practice, the attention paid to examining the EOL care patterns has been fairly limited.4 Therefore, my dissertation research is both needed and timely since it will fill this knowledge gap.
What have we known thus far?
Many studies primarily focused on the survival and treatment differences among individuals with and without dementia (including ADRD).21-26 Based on those studies, individuals with ADRD were commonly found to have delayed cancer diagnosis with unclear staging records.1,22,23 Also, they were more likely to have sub-optimal survival compared to their dementia-free counterparts.21-26 In terms of cancer treatment choices and care patterns, existing evidence indicated that older persons with cancer and dementia utilized diagnostic procedures, surgeries, chemotherapies, or radiation therapies at an early stage significantly lesser than the dementia-free group.22,23 One study group also found that individuals with dementias were unlikely to receive guideline-concordant cancer care compared with individuals without dementias.24
What evidence has been missing?
A few studies have deduced that the alarming relationship of having a dementia diagnosis in addition to cancer is associated with inferior survival and poorly planned treatment decisions.1,22,23,27 However, all of those assessments or speculations stated prior were derived based on the early 2000s healthcare claims data. As for assessing the economic characteristics of EOL care, previous healthcare service studies primarily focused on cost in relation to certain cancer types while ADRD was not considered as a separate independent factor in those estimations.27-30 Thus, the assessment of EOL care expenditures for older persons with cancer and ADRD is missing from the existing literature.
Therefore, the lack of pertinent research with respect to the substantial patient population (co-presence of cancer and ADRD) increase has become a knowledge gap in advancing patient care.2,28 My dissertation research filled this gap by assessing EOL care among older Medicare beneficiaries with breast-female only, lung, prostate, or colorectal cancer. My research answered two major questions: (1) What are the characteristics of aggressive EOL care utilization patterns in Medicare beneficiaries with both cancer and ADRD compared with those with just cancer? (2) Is there any EOL care cost difference among Medicare beneficiaries with cancer and ADRD compared with their non-ADRD counterparts?
How did I explore this?
To be consistent with current healthcare service research, my dissertation conducted two retrospective cohort studies (N Cohort I=135,380; N Cohort II=86,396) to assess the patterns of aggressive EOL care and estimating the Medicare expenditures of EOL care among beneficiaries (older than 65) with cancer and ADRD. I took advantage of the rich healthcare utilization record and cancer-related characteristics featured in the Surveillance, Epidemiology, End Results-Medicare (SEER-Medicare) database from 2004 to 2016.
To evaluate the EOL care patterns among beneficiaries with cancer and ADRD, I adopted six indicators to assess the aggressiveness of EOL care.29-31 The six specific factors are: (1) more than one ER visit during the last 30 days of life, (2) more than one inpatient admission during the last 30 days of life, (3) at least one ICU stay during the last 30 days of life, (4) death during inpatient admission, (5) initiation of hospice within 3 days of life, and (6) receiving any chemotherapy within 14 days of life.
For estimating the EOL spending paid by Medicare, the service dates and the payment from Medicare were captured from the following claims files: inpatient, outpatient, provider, hospice, home health care, and durable medical equipment.32-37 The spending of those different types of claims were summarized into: (1) monthly Medicare spending during the twelve months EOL period, (2) the total EOL Medicare spending of the twelve-month period, and (3) the total expenditures of each service type (i.e., inpatient, skilled nursing facility, outpatient, home health, durable medical equipment, and hospice) for the last twelve months.32-39
What did I find?
From 2004 to 2016, at least half of Medicare beneficiaries who had cancer had received a form of aggressive EOL care. In comparison with beneficiaries with cancer only, older persons with both ADRD and cancer had a slightly lower percentage of receiving aggressive EOL care throughout the years (Figure 2). From the multivariable logistic regression model, having ADRD attenuated older persons with cancer’s odds of utilizing any aggressive EOL care compared to those without ADRD.

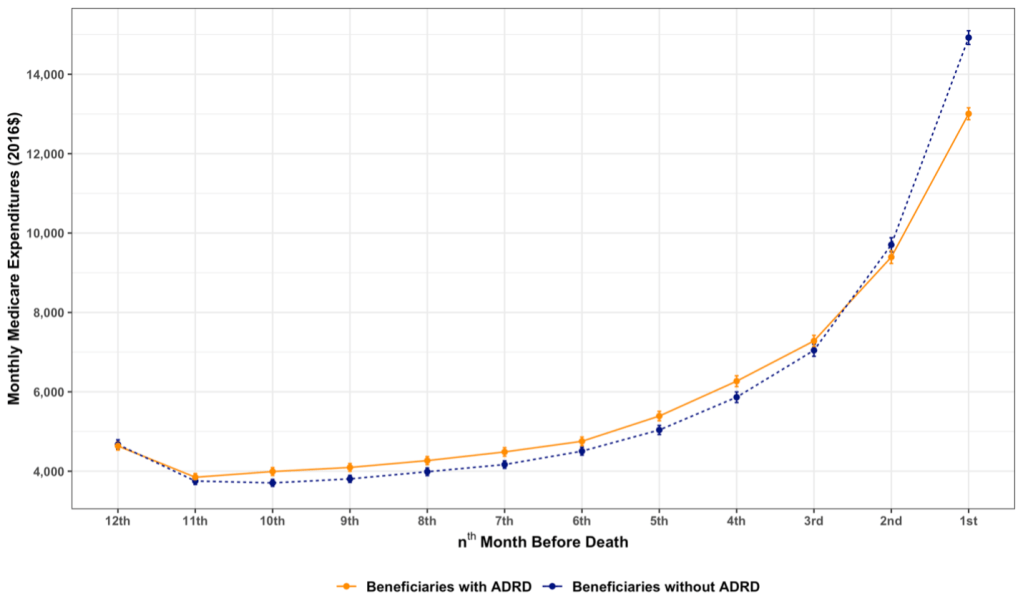
When examining the cost of EOL care, I found the majority of the expenditures incurred within the last six month of life (Figure 3). In comparison to those without ADRD, beneficiaries with both cancer and ADRD resulted in having slightly higher Medicare expenditures during the last twelve-month of life ($64,883 vs. $64,521). Specifically, greater expenditure was incurred on inpatient (6%), skilled nursing facility (126%), home health (42%), and hospice (24%) care among beneficiaries with both conditions in contrast with those without ADRD. After estimating the expenditure through statistical models (Generalized Estimating Equation Model and Generalized Linear Model), it was revealed cancer patients with ADRD had higher monthly expenditure, which resulted in higher in total expenditures.
Where will the findings lead us in public health and clinical care?
Featured studies from my dissertation revealed a relatively recent aggressive EOL care pattern and the financial burden for Medicare to pay for the EOL care among beneficiaries with both cancer and ADRD compared with those who did not have ADRD.
Two major future research directions could be carried out to further expand my dissertation’s impact in public health and clinical care. First, individuals diagnosed with subsequent ADRD after cancer have a higher risk of utilizing aggressive EOL care and spend more Medicare resources. Future studies could account for the sequence of ADRD and cancer diagnoses; then, evaluate its impact on EOL care patterns. Second, due to the limitation of healthcare claims data, future studies could deploy a mixed-method approach that combines both quantitative and qualitative data (e.g., family member’s interview, healthcare providers’ progress notes, palliative care consultation notes, and other EOL care program documents, etc.) together to evaluate the overall quality of EOL care.
Through conducting the proposed future studies in the field of EOL care, I am optimistic and confident that more health service researchers and clinicians will join this changing effort to advance EOL care quality while racing to find the cure of ADRD.
TL:DR
- Little is known about the quality of End-of-Life among cancer patients with Alzheimer’s Disease and Related Dementias (ADRD).
- From my study, cancer patients with ADRD utilized aggressive EOL care less; however, they tended to spend slightly more Medicare resources within the last twelve-month of life.
- Future efforts are needed to properly control the relative high prevalence of aggressive EOL care among older cancer patients and reduce the risk of the co-presence of cancer and ADRD on exacerbating the financial burden on Medicare.
References
1. Raji MA, Kuo YF, Freeman JL, Goodwin JS. Effect of a dementia diagnosis on survival of older patients after a diagnosis of breast, colon, or prostate cancer: implications for cancer care. Arch Intern Med. Oct 13 2008;168(18):2033-40. doi:10.1001/archinte.168.18.2033
2. Roderburg C, Loosen SH, Kunstein A, et al. Cancer Patients Have an Increased Incidence of Dementia: A Retrospective Cohort Study of 185,736 Outpatients in Germany. Cancers (Basel). Apr 22 2021;13(9)doi:10.3390/cancers13092027
3. National Cancer Institute. end-of-life care. January 20th, 2022. Accessed February 5th, 2022. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/end-of-life-care
4. Institute of Medicine. Dying in America: Improving quality and honoring individual preferences near the end of life. The National Academies Press; 2015.
5. National Cancer Institute. End-of-Life Care for People Who Have Cancer. Accessed January 20th, 2022. https://www.cancer.gov/about-cancer/advanced-cancer/care-choices/care-fact-sheet
6. Lee DH. Approach to end of life care. Ochsner J. Spring 2002;4(2):98-103.
7. Kaldjian LC, Curtis AE, Shinkunas LA, Cannon KT. Goals of care toward the end of life: a structured literature review. Am J Hosp Palliat Care. Dec-2009 Jan 2008;25(6):501-11. doi:10.1177/1049909108328256
8. Wright AA, Zhang B, Ray A, et al. Associations between end-of-life discussions, patient mental health, medical care near death, and caregiver bereavement adjustment. JAMA. Oct 8 2008;300(14):1665-73. doi:10.1001/jama.300.14.1665
9. Wright AA, Keating NL, Balboni TA, Matulonis UA, Block SD, Prigerson HG. Place of death: correlations with quality of life of patients with cancer and predictors of bereaved caregivers’ mental health. J Clin Oncol. Oct 10 2010;28(29):4457-64. doi:10.1200/JCO.2009.26.3863
10. Lin CP, Cheng SY, Chen PJ. Advance Care Planning for Older People with Cancer and Its Implications in Asia: Highlighting the Mental Capacity and Relational Autonomy. Geriatrics (Basel). Jul 20 2018;3(3)doi:10.3390/geriatrics3030043
11. Sarfati D, Koczwara B, Jackson C. The impact of comorbidity on cancer and its treatment. CA Cancer J Clin. Jul 2016;66(4):337-50. doi:10.3322/caac.21342
12. Alzheimer’s Association. 2020 Alzheimer’s disease facts and figures. Alzheimer’s & dementia : the journal of the Alzheimer’s Association. Mar 10 2020;doi:10.1002/alz.12068
13. Schachter AS, Davis KL. Alzheimer’s disease. Dialogues Clin Neurosci. Jun 2000;2(2):91-100.
14. National Institute on Aging. Alzheimer’s Disease Fact Sheet. Accessed February 3rd, 2022. https://www.nia.nih.gov/health/alzheimers-disease-fact-sheet
15. White MC, Holman DM, Boehm JE, Peipins LA, Grossman M, Henley SJ. Age and cancer risk: a potentially modifiable relationship. Am J Prev Med. Mar 2014;46(3 Suppl 1):S7-15. doi:10.1016/j.amepre.2013.10.029
16. Smith BD, Smith GL, Hurria A, Hortobagyi GN, Buchholz TA. Future of cancer incidence in the United States: burdens upon an aging, changing nation. J Clin Oncol. Jun 10 2009;27(17):2758-65. doi:10.1200/JCO.2008.20.8983
17. Hopkinson JB, Milton R, King A, Edwards D. People with dementia: what is known about their experience of cancer treatment and cancer treatment outcomes? A systematic review. Psychooncology. Oct 2016;25(10):1137-1146. doi:10.1002/pon.4185
18. Husebo BS, Strand LI, Moe-Nilssen R, Husebo SB, Snow AL, Ljunggren AE. Mobilization-Observation-Behavior-Intensity-Dementia Pain Scale (MOBID): development and validation of a nurse-administered pain assessment tool for use in dementia. J Pain Symptom Manage. Jul 2007;34(1):67-80. doi:10.1016/j.jpainsymman.2006.10.016
19. Coleman AM. End-of-life issues in caring for patients with dementia: the case for palliative care in management of terminal dementia. Am J Hosp Palliat Care. Feb 2012;29(1):9-12. doi:10.1177/1049909111410306
20. National Cancer Institute. NCI Funded Research Protfolio. Accessed 2022, March 10th. https://fundedresearch.cancer.gov/nciportfolio/search/SearchForm
21. Baillargeon J, Kuo YF, Lin YL, Raji MA, Singh A, Goodwin JS. Effect of mental disorders on diagnosis, treatment, and survival of older adults with colon cancer. J Am Geriatr Soc. Jul 2011;59(7):1268-73. doi:10.1111/j.1532-5415.2011.03481.x
22. Gorin SS, Heck JE, Albert S, Hershman D. Treatment for breast cancer in patients with Alzheimer’s disease. J Am Geriatr Soc. Nov 2005;53(11):1897-904. doi:10.1111/j.1532-5415.2005.00467.x
23. Gupta SK, Lamont EB. Patterns of presentation, diagnosis, and treatment in older patients with colon cancer and comorbid dementia. J Am Geriatr Soc. Oct 2004;52(10):1681-7. doi:10.1111/j.1532-5415.2004.52461.x
24. Kimmick G, Fleming ST, Sabatino SA, et al. Comorbidity burden and guideline-concordant care for breast cancer. J Am Geriatr Soc. Mar 2014;62(3):482-8. doi:10.1111/jgs.12687
25. Centers for Disease Control and Prevention. Cancer Survival in the United States. Accessed September 15th, 2020. https://www.cdc.gov/cancer/dcpc/research/articles/concord-2-supplement.htm
26. Patnaik JL, Byers T, Diguiseppi C, Denberg TD, Dabelea D. The influence of comorbidities on overall survival among older women diagnosed with breast cancer. J Natl Cancer Inst. Jul 20 2011;103(14):1101-11. doi:10.1093/jnci/djr188
27. Raji MA, Tamborello LP, Kuo YF, et al. Risk of subsequent dementia diagnoses does not vary by types of adjuvant chemotherapy in older women with breast cancer. Med Oncol. Dec 2009;26(4):452-9. doi:10.1007/s12032-008-9145-0
28. McWilliams L, Farrell C, Grande G, Keady J, Swarbrick C, Yorke J. A systematic review of the prevalence of comorbid cancer and dementia and its implications for cancer-related care. Aging Ment Health. Oct 2018;22(10):1254-1271. doi:10.1080/13607863.2017.1348476
29. Earle CC, Park ER, Lai B, Weeks JC, Ayanian JZ, Block S. Identifying potential indicators of the quality of end-of-life cancer care from administrative data. J Clin Oncol. Mar 15 2003;21(6):1133-8. doi:10.1200/JCO.2003.03.059
30. Earle CC, Neville BA, Landrum MB, et al. Evaluating claims-based indicators of the intensity of end-of-life cancer care. Int J Qual Health Care. Dec 2005;17(6):505-9. doi:10.1093/intqhc/mzi061
31. Karanth S, Rajan SS, Revere FL, Sharma G. Factors Affecting Racial Disparities in End-of-Life Care Costs Among Lung Cancer Patients: A SEER-Medicare-based Study. Am J Clin Oncol. Feb 2019;42(2):143-153. doi:10.1097/COC.0000000000000485
32. Centers for Medicare and Medicaid Services. Medicare costs at a glance. Accessed October 21st, 2020. https://www.medicare.gov/your-medicare-costs/medicare-costs-at-a-glance
33. ResDAC. Line Beneficiary Coinsurance Amount. Accessed October 21st, 2020. https://www.resdac.org/cms-data/variables/line-beneficiary-coinsurance-amount
34. ResDAC. Claim (Medicare) Payment Amount. Accessed October 21st, 2020. https://www.resdac.org/cms-data/variables/claim-medicare-payment-amount
35. ResDAC. NCH Beneficiary Part B Deductible Amount. Accessed October 21st, 2020. https://www.resdac.org/cms-data/variables/nch-beneficiary-part-b-deductible-amount
36. ResDAC. MEDPAR Beneficiary Inpatient Deductible Liability Amount. Accessed October 21st, 2020. https://www.resdac.org/cms-data/variables/medpar-beneficiary-inpatient-deductible-liability-amount
37. ResDAC. NCH Beneficiary Inpatient (or other Part A) Deductible Amount. Accessed October 21st, 2020. https://www.resdac.org/cms-data/variables/nch-beneficiary-inpatient-or-other-part-deductible-amount
38. Chronic Condition Data Warehouse. CCW Technical Guidance: Getting Started with CMS Medicare Administrative Research Files. Accessed October 11th, 2021. https://www2.ccwdata.org/documents/10280/19002248/ccw-technical-guidance-getting-started-with-cms-medicare-administrative-research-files.pdf
39. Bremner KE, Yabroff KR, Coughlan D, et al. Patterns of Care and Costs for Older Patients With Colorectal Cancer at the End of Life: Descriptive Study of the United States and Canada. JCO Oncol Pract. Jan 2020;16(1):e1-e18. doi:10.1200/JOP.19.00061