By Victoria Silvis
Since the first successful kidney transplant in 1954, organ transplantation has become an increasingly common procedure with over 39,000 occurring in the United States in 2020.1,2 While the pancreas, heart, and liver were the next organs to be transplanted in the 1960’s, organ procurement was a challenge, as the United Network for Organ Sharing (UNOS) was not formed until 1977.1 UNOS is a computerized network used to match organs with transplant patients that considers only medical and logistical factors while avoiding socioeconomic status and insurance coverage factors.3 Before UNOS, physicians everywhere, even those here at Penn State, got creative to help save lives of numerous patients while waiting for a potential organ match in the local area.
Open-heart surgery includes any surgery requiring the chest to be cut open. These surgeries most commonly repair or replace damaged heart valves and arteries, implant a pacemaker or similar assist device, or insert a heart transplant.4 The heart must be immobile to perform surgical tasks on it, but blood flow is crucial to the patient. To ensure oxygenated blood flows through the body during surgery, the patient is placed on a heart-lung machine, also known as a cardiopulmonary bypass machine (CPB). CPB is connected to the right atrium, where deoxygenated blood would typically enter the heart, and the aorta, the main artery to pump oxygenated blood to the body (Figure 1). Deoxygenated blood is moved from the right atrium to an oxygenator to provide oxygen to the red blood cells. Once oxygenated, the blood can move directly to the rest of the body via the aorta, allowing blood to skip the heart and lungs entirely.5 Patients can be connected to CPB for hours throughout the surgery, but with increased time on CPB, mortality and morbidity rates increase.6 This is likely due to a potential increase in surgical site infections.7
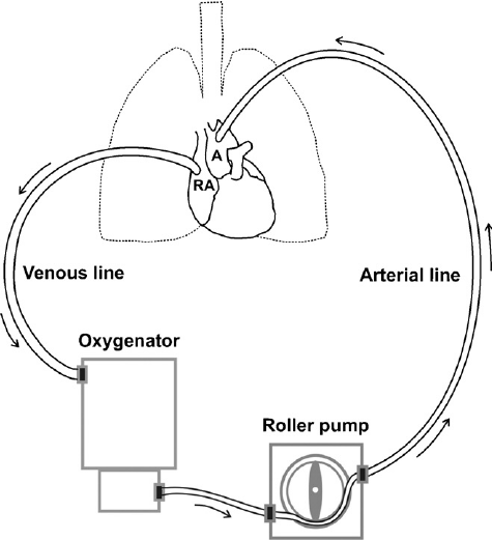
Patients must be carefully and gradually weaned off of CPB following surgery. Weaning is successful when heart rate, mean arterial pressure, ventricle filling, and cardiac index are stable and within specified parameters. Some patients are unable to be fully weaned off of CPB due to vasodilation, bleeding, or inadequate perfusion of the heart. Patients who require heart surgery often have a weakened heart to begin with and surgery weakens it further, meaning the extra support provided by CPB may be needed still.9 About 1-5% of patients cannot be weaned from CPB and cannot immediately get a new heart to replace the diseased one.10 Patients who cannot be weaned or whose heart is too weak to sustain them until a donor can be found have benefited immensely from the Penn State Heart-Assist Pump.
The Penn State Heart-Assist Pump (PSHAP) also known as the Pierce-Donachy ventricular assist device began development in the 1970’s. Cardiothoracic surgeon, Dr. William Pierce, and fabrications specialist, James Donachy were the two main developers and were assisted by two Penn State mechanical engineers, John Brighton and Winfred Phillips, and mechanical engineering graduate student, Gerson Rosenberg.11 Multiple prototypes of this artificial heart and pump were created before settling on the final device patented in 1980, the same year it received FDA approval. This device was revolutionary as it was the first heart assist pump to be surgically implanted into a patient.10
The PSHAP was used primarily as a bridge device to sustain patients for weeks to months before transplant. Originally, the PSHAP was designed to bypass the left ventricle of the heart by connecting to the left atrium and aorta10 (Figure 2). The left ventricle pumps oxygenated blood through the aorta to the rest of the body and loss of left ventricular function is the most common cause of heart failure.13 The chest of the patient could remain closed while the tubes connecting the pump and heart entered through small abdominal incisions with the pump itself rested on the outside of the abdomen10 (Figure 3).

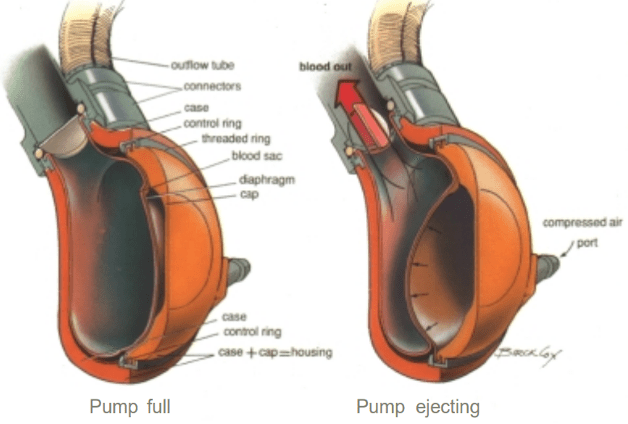
The pumping mechanism of the PSHAP utilizes a pneumatic drive unit which transforms pressurized air into energy and in turn, drives rotational movement of the blood.10,14 However, air acts as a potential hazard because air bubbles in the bloodstream can lead to an air embolism, block blood flow, and lead to a stroke or heart attack.15 To combat the threats of an air embolism or a blood clot, the sac housing the blood is smooth and seamless to prevent it from rupturing. Further, the plastic case that houses the device is also incredibly smooth to alleviate potential snags. The plastic housing is comprised of a main case and a cap which contains an inflatable diaphragm. The cap and diaphragm provide the final failsafe of the pump as the pressurized air moves the diaphragm away from the cap, pushing on the blood-containing sac to pump blood back to the aorta. During this movement, the walls of the sac do not come into contact and if the blood sac ruptures, the diaphragm keeps air away from the tubing connected to the heart10 (Figure 4). Although it is no longer used, a total of 55 patients received a PSHAP at Hershey Medical Center and over 200 patients received a PSHAP worldwide with zero fatalities resulting from device failure.10
However, this isn’t where the ingenuity of the PSHAP stops. Anthony Mandia, a 44-year-old Philadelphia resident, was transferred to Hershey Medical Center on October 14, 1985, suffering from an enlarged and failing heart, or cardiomegaly. He was immediately placed on the heart transplant list after discovering the urgency of his condition. Mandia’s heart was almost twice the size of a normal, healthy heart. Only three days after entering the hospital and after trying multiple methods of circulatory support with no success, physicians proposed implanting an artificial heart into Mandia.16 On the morning of October 18th, 1985, Mandia received two modified PSHAPs allowing them to serve as a total artificial Penn State Heart (PSH), (Figure 5) affording Mandia time to receive a donor heart. Mandia was one of three patients to receive the PSH as a total artificial heart, but luckily, he only needed it for 10 days before a transplant was found. A second patient who received the total artificial PSH survived on it for 390 days before a donor was found!10

The Penn State Heart-Assist Pump and Penn State Heart served as building blocks for future artificial heart development, and I encourage you to go visit this revolutionary device in-person in various entryways to the hospital. There are many innovative ways patients on the transplant waitlist can have improved quality of life while waiting for a donor, and there are many ways that you can help people in need of various transplants. Consider becoming a living donor where you can donate blood, bone marrow, or even kidneys and parts of your liver. With over 100,000 people waiting on a transplant, only about a third receive the donation they need to survive. Imagine how many lives we could save if more people become a registered organ and tissue donor. Organ donors can save up to 8 lives and this increases to 75 lives with tissue donations!18
TL:DR
- Heart-Assist Pump created at PSMSHMC revolutionized artificial heart development.
- While bridge devices aid patients to transplant, organ donors are needed.
References
- https://unos.org/transplant/history/
- https://www.organdonor.gov/learn/organ-donation-statistics
- https://unos.org/transplant/how-we-match-organs/
- https://www.healthline.com/health/open-heart-surgery#when-it’s-needed
- https://www.texasheart.org/heart-health/heart-information-center/topics/a-heart-surgery-overview/
- https://www.peertechzpublications.com/articles/AC-2-104.php
- https://www.sciencedirect.com/science/article/pii/S2405857219300816
- https://www.researchgate.net/publication/24424433_Pulmonary_blood_flow_generates_cardiogenic_oscillations
- https://www.uptodate.com/contents/weaning-from-cardiopulmonary-bypass
- https://www.asme.org/wwwasmeorg/media/resourcefiles/aboutasme/who%20we%20are/engineering%20history/landmarks/142-pierce-donachy-ventricular-assist-device.pdf
- https://research.med.psu.edu/departments/heart-vascular-institute/innovation/
- https://moosmosis.org/2020/07/09/circulatory-system-blood-flow-pathway-through-the-heart/
- https://www.heart.org/en/health-topics/heart-failure/what-is-heart-failure/types-of-heart-failure
- https://www.tme.eu/en/news/library-articles/page/43313/industrial-pneumatic-drives/#:~:text=Pneumatic%20drives%20operate%20based%20on,into%20linear%20or%20rotational%20movement.
- https://www.nhs.uk/conditions/air-embolism/#:~:text=Air%20can%20escape%20from%20the,a%20stroke%20or%20heart%20attack
- https://www.collegian.psu.edu/archives/chronology-of-mandia-and-the-penn-state-heart/article_bc547d9c-ebd2-52b3-8076-21372b6371a3.html
- https://www.jtcvs.org/article/S0022-5223(19)35815-5/pdf
- https://www.donors1.org/learn-about-organ-donation/who-can-donate/get-the-facts/
