By Ian Hayman
Bird Flu. Ebola. Hantavirus. Dengue. Zika. SARS-CoV2. Bubonic Plague. West Nile. Malaria. These pathogens are often cast as the central antagonist, the great evil in many books, movies, and in real life. Even hearing their names can produce a visceral response, a sense of dread in the back of the head about these bogeymen of the microbial world. All of these infectious agents have one thing in common: they are zoonotic and vector-borne diseases, spread by and maintained in non-human animal or insect reservoirs, respectively. The ability for a pathogen to jump between species means that these diseases can abruptly emerge into the human population. Simply treating humans as they become infected will not eliminate the threat posed by animal and insect borne diseases. So long as non-human reservoirs exist, and humans come into contact with them, there will be a persistent risk of emerging disease.

Over 60% of today’s emerging infectious diseases are of zoonotic origin, and over 70% of zoonotic diseases originate from wild animal populations1. Insect vector-borne illnesses account for over 28% of all emerging disease events in the past decade1. Together, just shy of 90% of all new emerging diseases come from animals and insects interacting with human populations1. Unexpected jumps of emerging pathogens into the human population can have devastating effects, as seen with Bubonic Plague (Fig 1). Genetic surveillance of circulating pathogens in animal and insect populations can identify the presence of dangerous pathogens before they enter human populations2. There are two strategies currently employed to prevent identified emerging infectious diseases from jumping to humans: 1) removal of contact between humans and vectors or animal reservoirs, and 2) eliminating the insect vector or animal population2; 3.
Genetic Modification of Insect Vectors
Mosquitos serve as the insect vectors for numerous important human pathogens, including Dengue, Zika, West Nile, and Malaria. Mosquito nets are an example of the first disease control strategy, and effectively reduce the incidence of disease in human populations by removing points of contact between humans and disease vectors. Pesticide spraying and insect traps are examples of the second control strategy that focuses on eliminating the population of disease vectors. Both strategies are effective at reducing rates of infection in humans, but they also carry significant implementation problems4. Many zoonotic and vector-borne diseases occur in tropical areas and in the developing world, which makes it difficult to transport and deploy individualized protection measures to a large enough percentage of the population to prevent transmission4. Additionally, the use of pesticides and insect traps negatively impacts beneficial insect populations, such as critical pollinators, although ultra-low volume application can reduce these impacts5; 6; 7. Because of these challenges, more specific and targeted techniques are required to effectively eliminate the threat of emerging infectious diseases.
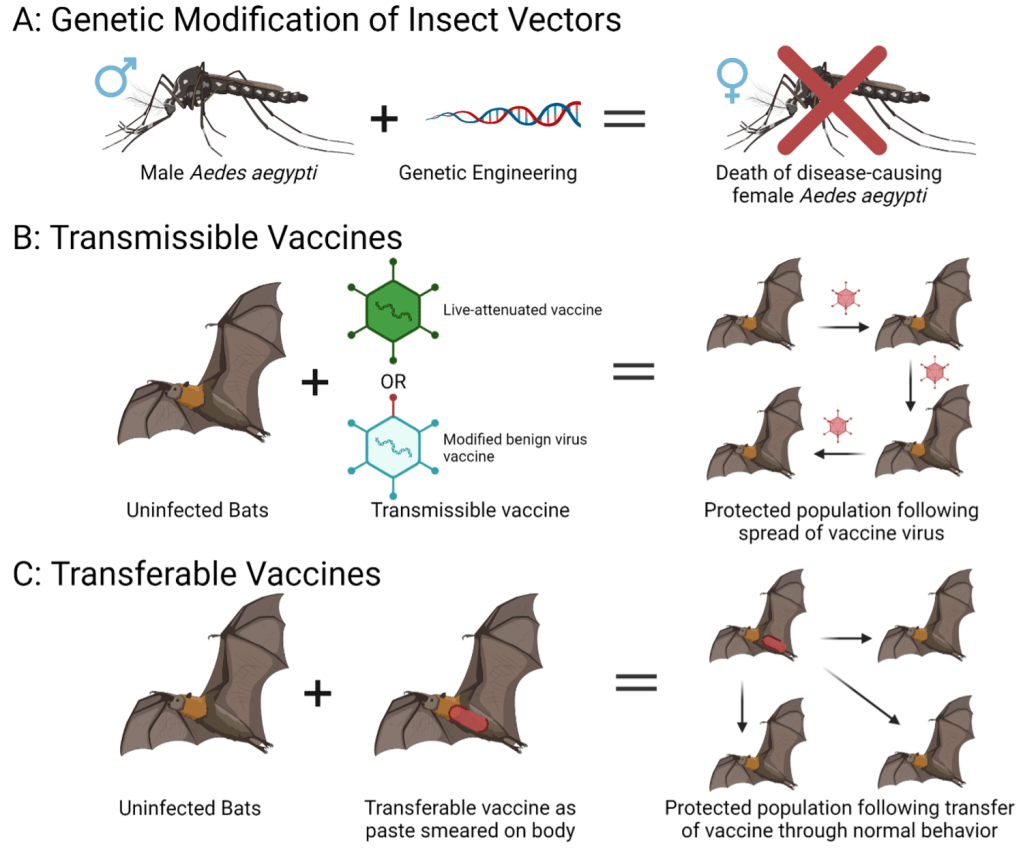
One of these more targeted techniques was deployed in April of this year: the first genetically modified mosquitos, designed to reduce mosquito-borne illnesses, were released in Florida8. While the Aedes aegypti mosquito represents only 4% of the total mosquito population in Florida, this mosquito is responsible for essentially all mosquito-borne illness in the state8. The biotech firm Oxitec developed genetically modified male mosquitos that pass a gene onto their offspring that kills all female larvae8. A mosquito must bite a human to transmit a mosquito-borne illness, and only female mosquitos bite humans8. Oxitec’s approach aims to both reduce disease transmission, by specifically reducing the female mosquito vector, while also functionally eliminating the Aedes aegypti mosquito population due to a lack of females for breeding.
Vaccinating Animal Reservoirs of Zoonotic Disease
Beyond more targeted extermination efforts, some scientists have begun applying vaccination strategies to disease reservoirs9; 10; 11. Vaccination programs are highly suited for prevention of disease in the animals that serve as disease reservoirs, including avian and mammalian species9. Vaccination efforts applied to domesticated animal populations benefit from the fact that these animals are already contained, thus it is easier to ensure complete coverage of treatment. In contrast, difficulties arise with wild animal populations, where vaccination strategies must account for the inability to directly vaccinate all individuals. Therefore, self-disseminating animal vaccines may be the best strategy for the elimination of zoonotic diseases before they even reach the human population9; 10; 11.
Self-disseminating vaccines currently utilize two different mechanisms of spread:
1) transmissible vaccines that infect and spread within an animal population like a normal pathogen, undergoing multiple transmission events between animals, and 2) transferable vaccines that can be applied to a single animal and undergo only a single transmission event to other animals in that population (Fig 2)9.
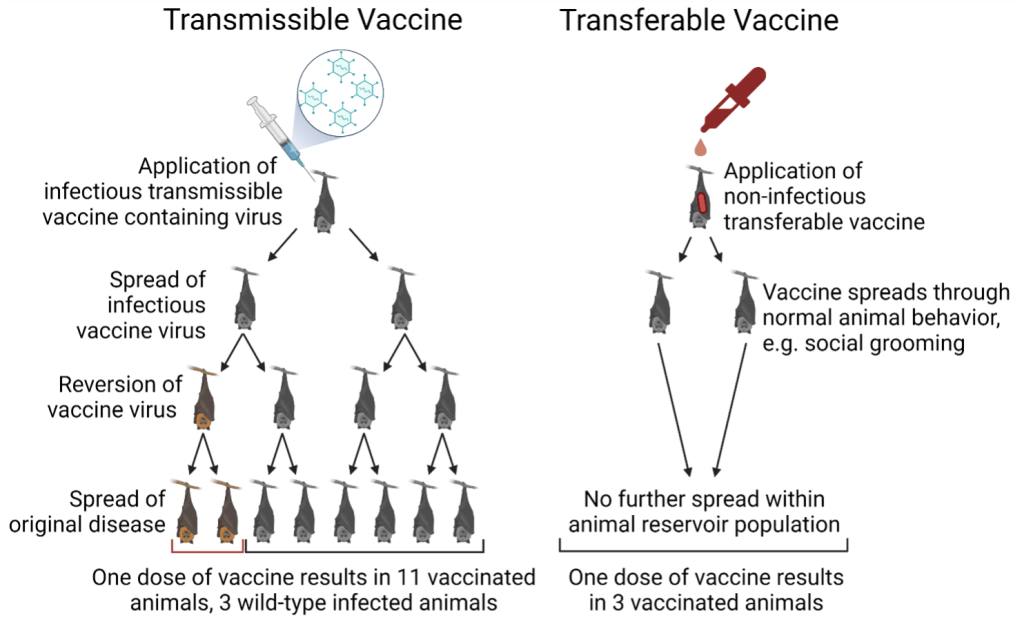
Transmissible vaccines require a benign pathogen at their core. A transmissible live attenuated vaccine utilizes a weakened version of the pathogen targeted by the vaccine, eliciting an immune response without causing adverse symptoms. Unfortunately, the weakened viruses at the core of live attenuated vaccines can mutate back to a more dangerous wild-type pathogen, known as a revertant12. Vaccine reversion is a documented phenomenon that happens with the live attenuated polio vaccine, and while vaccinated individuals retain protection against revertants, they can pass functional polio virus in their stool and spread the disease to unvaccinated individuals12. The current leading cause of Type 2 polio infection is the vaccine itself; most outbreaks that occur today arise from the vaccine strain of poliovirus spreading in unvaccinated populations12.
One method to avoid problems arising from live attenuated vaccines involves genetic recombineering13. A benign virus, such as adenovirus, can be genetically modified to include a specific protein from a pathogen you want to vaccinate against. Because there is a wild-type benign virus at the core of these vaccines, they can’t revert to a pathogenic strain. If genetic mutations cause the added pathogen protein to be lost, you are just left with the normal benign virus. However, the continued spread of the benign virus at the core of your vaccine would still be problematic because the target animal population would develop immunity against the benign virus without developing protection against your pathogen of interest. This prevents the use of this benign virus in developing future vaccines, but that is preferable to the spread of a dangerous pathogen.
Unlike transmissible vaccines, transferable vaccines avoid the issues discussed above by limiting spread to a single transmission event. Transferable vaccines take advantage of animal behavior. For example, bats are a vector for several zoonotic diseases of concern, including Ebola and Nipah virus14; 15. Vaccines developed as a paste and smeared on the back of bats can serve as transmissible vaccines. When the bat grooms itself, it will ingest some of the vaccine and gain protection against the pathogen of interest. Bats also groom each other, so every bat that grooms the bat smeared with vaccine paste will also ingest the vaccine and become protected9. There is no infectious virus within the transferable vaccine, so there is no further spread of the vaccine beyond this initial physical transfer resulting from grooming9. Such vaccines can be disseminated within an animal reservoir without needing to capture and vaccinate each individual animal and don’t carry the risk of a reverting pathogen as there is no infectious virus present. Overall spread of the vaccine within the population would be slower, however, meaning that the spread of the wild-type pathogen targeted by the vaccine could potentially outpace and nullify the benefits of the vaccine9; 10.
Conclusion and Closing Thoughts
Controlling pathogen reservoirs within animal and insect vectors will be required for the effective eradication of emerging infectious diseases. Many of the emerging zoonotic and vector-borne diseases are endemic to tropical climates and as climate change accelerates, it is likely that the insect and animal hosts for these diseases will gain new territory in regions currently unaffected by them16. Emerging diseases are currently controlled by either eliminating points of contact between disease reservoirs and humans, or by eliminating the populations of disease reservoirs. New technologies allow us to target zoonotic and vector-borne diseases in new ways: genetic modification of male mosquitos may allow for targeted destruction of reservoir populations without impacting other insect populations, and self-disseminating vaccines may allow us to eliminate pathogen reservoirs in animals without killing the animals themselves (Fig 3).
Self-disseminating vaccine technologies may be available in the future for humans as well, which raises numerous ethical questions. If self-disseminating vaccines can eliminate pathogens within non-human animal populations, could they be used to rapidly vaccinate human populations against emerging pathogens? Could this technology eliminate the need for rich nations to horde vaccine doses while poorer nations suffer, as currently seen with COVID-19 vaccines? Is it ethical for people to be vaccinated, possibly against their will or knowledge, due to a self-disseminating vaccine? These are questions for the future, but hopefully a future that is not too distant.
References
1 KE, J. et al. Global trends in emerging infectious diseases. Nature, v. 451, n. 7181, 02/21/2008 2008. ISSN 1476-4687. < https://www.ncbi.nlm.nih.gov/pubmed/18288193 >.
2 GARY GARDNER, T. P., MICHAEL RENNER, KATIE AUTH, BEN CALDECOTT, PETER DASZAK, HEATHER EXNER-PIROT, GARY GARDNER, FRANÇOIS GEMENNE, NATHAN JOHN HAGENS, TIM JACKSON, WILLIAM B. KARESH, ELIZABETH H. LOH, CATHERINE C. MACHALABA, TOM PRUGH, ROBERT RAPIER, MICHAEL RENNER, AND PETER A. VICTOR. Emerging Diseases from Animals. Nature Public Health Emergency Collection. 2015
3 SS, M. et al. Prediction and prevention of the next pandemic zoonosis. Lancet (London, England), v. 380, n. 9857, 12/01/2012 2012. ISSN 1474-547X. < https://www.ncbi.nlm.nih.gov/pubmed/23200504 >.
4 DAHMANA, H.; MEDIANNIKOV, O. Mosquito-Borne Diseases Emergence/Resurgence and How to Effectively Control It Biologically. Pathogens, v. 9, n. 4, 2020. < http://dx.doi.org/ >.
5 H, Z. et al. Minimizing the impact of the mosquito adulticide naled on honey bees, Apis mellifera (Hymenoptera: Apidae): aerial ultra-low-volume application using a high-pressure nozzle system. Journal of economic entomology, v. 97, n. 1, 2004 Feb 2004. ISSN 0022-0493. < https://www.ncbi.nlm.nih.gov/pubmed/14998120 >.
6 WA, Q.; RD, X.; H, Z. Impact of bifenthrin on honeybees and Culex quinquefasciatus. Journal of the American Mosquito Control Association, v. 26, n. 2, 2010 Jun 2010. ISSN 8756-971X. < https://www.ncbi.nlm.nih.gov/pubmed/20649134 >.
7 A, C. et al. Nontarget effects of aerial mosquito adulticiding with water-based unsynergized pyrethroids on honey bees and other beneficial insects in an agricultural ecosystem of north Greece. Journal of medical entomology, v. 51, n. 3, 2014 May 2014. ISSN 0022-2585. < https://www.ncbi.nlm.nih.gov/pubmed/24897869 >.
8 WALTZ, E. First genetically modified mosquitoes released in the United States. Nature: Nature Publishing Group. 593: 175-176 p. 2021.
9 NUISMER, S. L.; BULL, J. J. Self-disseminating vaccines to suppress zoonoses. Nature Ecology & Evolution, v. 4, n. 9, p. 1168-1173, 2020-07-27 2020. ISSN 2397-334X2397-334X. < https://www.nature.com/articles/s41559-020-1254-y?fbclid=IwAR2WEQzvQtjh6M8QoTtdGiWpzY4OA5abVlWj6VYsDK0gTedR9REBD92vqfc#ref-CR20 >.
10 JJ, B.; MW, S.; SL, N. Transmissible Viral Vaccines. Trends in microbiology, v. 26, n. 1, 2018 Jan 2018. ISSN 1878-4380. < https://www.ncbi.nlm.nih.gov/pubmed/29033339 >.
11 CL, S.; SL, N.; AJ, B. When to vaccinate a fluctuating wildlife population: Is timing everything? The Journal of applied ecology, v. 57, n. 2, 2020 Feb 2020. ISSN 0021-8901. < https://www.ncbi.nlm.nih.gov/pubmed/32139945 >.
12 AL, V. et al. The Early Evolution of Oral Poliovirus Vaccine Is Shaped by Strong Positive Selection and Tight Transmission Bottlenecks. Cell host & microbe, v. 29, n. 1, 01/13/2021 2021. ISSN 1934-6069. < https://www.ncbi.nlm.nih.gov/pubmed/33212020 >.
13 J, B. et al. Horizontal transmissible protection against myxomatosis and rabbit hemorrhagic disease by using a recombinant myxoma virus. Journal of virology, v. 74, n. 3, 2000 Feb 2000. ISSN 0022-538X. < https://www.ncbi.nlm.nih.gov/pubmed/10627521 >.
14 CALISHER, C. H. et al. Bats: Important Reservoir Hosts of Emerging Viruses. Clinical Microbiology Reviews, v. 19, n. 3, p. 531-545, 2006. < http://dx.doi.org/ >.
15 WYNNE, J. W.; WANG, L. F. Bats and Viruses: Friend or Foe? In: (Ed.). PLoS Pathog, v.9, 2013. ISBN 1553-7366 (Print)1553-7374 (Electronic).
16 EL-SAYED, A.; KAMEL, M. Climatic changes and their role in emergence and re-emergence of diseases. Environmental Science and Pollution Research, p. 1-17, 2020. < http://dx.doi.org/ >.