By Hannah Johnson

Have you ever wondered about the industry behind the color of your food? So much of what we eat is transformed into more vibrant and distinguishable colors and this is especially true for manufactured foods, such as candy, cereal, and jelly. A key benefit of food coloring is that it allows our sight to enhance the flavors we experience while eating food. Many synthetic and natural color additives are in use today to enrich our experiences of food, medicine, and cosmetics. Most of the synthetic dyes have a natural alternative; however, this isn’t the case for Blue No. 1 dye.
The history of synthetic color additives began in 1856 when British chemist, William Henry Perkin, discovered the first organic synthetic dye mauve1,4. Perkin had accidently discovered this dye, coined mauveine, while attempting to make a synthetic version of quinine (at the time, quinine was the treatment of choice for malaria)6. Perkin’s discovery prompted production of a vast collection of synthetic dyes. However, at the time there were no protective regulations in place, and thus manufacturers were able to sell almost anything they were able to produce. Specifically, some people actually used dyes to sell spoiled or defective food to customers and it was later found that many of these newfound synthetic color additives contained harmful materials such as lead, arsenic, and mercury1. In an effort to rectify this problem, the United States officially began federally regulating color additives in the 1880s in an effort to prevent people from consuming harmful products1. These findings further led to the establishment of the Food and Drug Act in 1906 prohibiting the use of color additives containing poisonous material or for use in hiding defective products1. It was the formation of the Federal Food, Drug, and Cosmetic (FD&C) Act in 1938, however, that really began the FDA’s standards for color additives.
The FD&C Act started the requirement for color additives to be officially certified, as well as included cosmetics and medications within their purview. At this point, there were only fifteen federally approved synthetic color additives. However, after unfortunate events on Halloween 1950, where candy containing FD&C-approved Orange No. 1 caused many children to become sick, the FD&C Act obtained an amendment. The 1960 amendment resulted in more stringent requirements for food color additives to be deemed “suitable and safe”1. This amendment resulted in the founding of the current seven well-known synthetic color additives, which are sometimes collectively known as the coal-tar colors1,2,4. These FD&C Act-approved color additives can be seen in Figure 1, along with a corresponding natural alternative to each synthetically produced color. The name “coal-tar colors” is attributed to their original production from coal by-products2. The coal-tar colors are now produced from petroleum with stringent testing undertaken to ensure nothing remains of the original petroleum in the final product1,2,4.
The seven FD&C Act-approved synthetic color additives have been largely used in food, medicine, and cosmetics for many years; however, there is a growing push for replacing artificial dyes with natural alternatives in food manufacturing4. Many food manufacturers such as Mars, Nestle, and Kraft established a shift in their use of natural food color additives in response to a 2010 European Union requirement to add the label of “may have an adverse effect on activity and attention in children” on food containing artificial dyes4. For many colors, there is an already established natural alternative. For example, margarine and cheese can obtain their orange tint through the addition of b-carotene, which can be found in sweet potatoes and pumpkins2. These natural color additives can fairly easily replace many synthetic dyes. However, the Blue No. 1 dye, also called Brilliant Blue, has no such natural alternative. An extensive search was conducted by these companies to find the elusive natural Brilliant Blue. Eventually, this search led to the discovery of spirulina, a form of algae containing a blue-green color3,4. The FDA approved spirulina in 2013 for use as a color additive, but the discovery of spirulina did not completely solve the problem. Spirulina wasn’t a perfect solution to Blue No. 1. It tended to give an aftertaste not seen in its synthetic counterpart Blue No. 1 and, most importantly, it was not capable of obtaining the correct hue needed to match Blue No. 14. A better alternative is still sorely sought after for many companies to truly turn away from synthetic color additives.
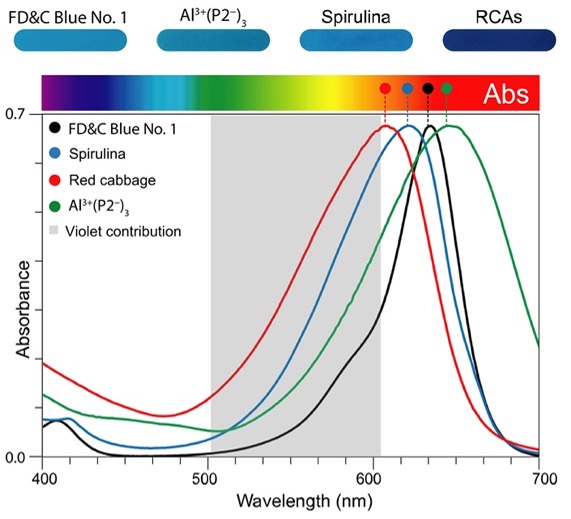
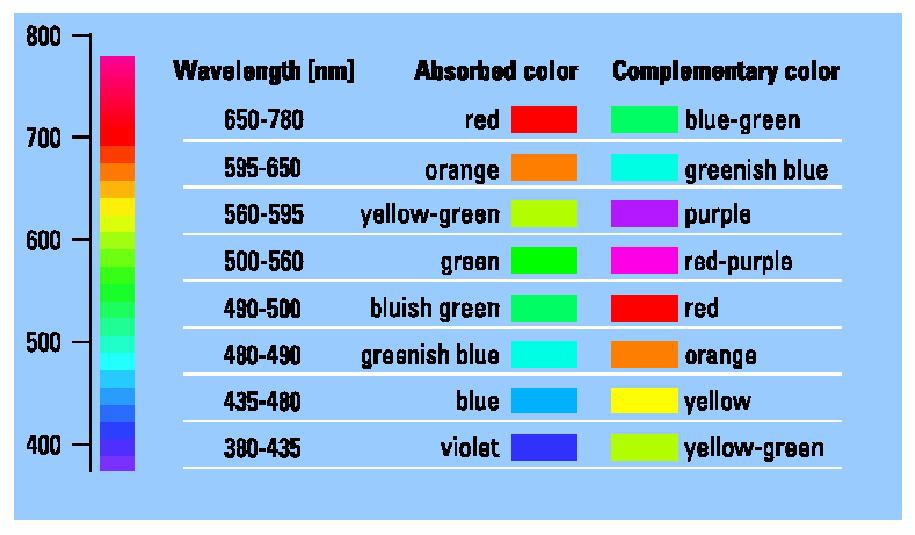
Recently, a study was published showcasing the discovery of an alternative pathway to produce a color matching the cyan blue hue of Blue No. 13. Denish et al. utilized anthocyanin, a compound found in red cabbage known for giving a deep purple or blue color3. They were able to identify a total of eight anthocyanin homologs within red cabbage designated as P1-P83. In order to determine if any of these anthocyanins might be used for a natural Blue No. 1 color additive, Denish et al. measured the absorbances of the P1-P8 anthocyanins to obtain the wavelength given at the highest point of absorption for each homolog. The use of wavelength and absorption to discuss color may seem counter intuitive. When light reflects or passes through something, the color perceived through our vision is actually the complimentary of the color absorbed (e.g., we see blue when orange is absorbed) (Figure 3). For example, as seen on the graph in Figure 2, the wavelength recorded for Blue No. 1 of 630nm corresponds to its highest measurement of absorption. Of the P1-P8 anthocyanins, Denish et al. were able to pinpoint that the P2 monoacetylated anthocyanin has a wavelength of 640 nm when at neutral pH3. Thus, P2 appears to be closer in wavelength to Blue No. 1 than spirulina, as Blue No 1. has a wavelength of 630nm and spirulina has a wavelength of 613 nm (Figure 2)3. As a result, spirulina is closer to having a violet tint than the greenish-blue coloring needed to match Blue No. 1 (Figure 3). On the other hand, the P2 anthocyanin is much closer to the wavelength of Blue No. 1 that is needed to achieve the necessary hue. However, as P2 only qualifies as less than 5% of the total anthocyanins contained in red cabbage, further research was required to uncover if any of the P1-P8 anthocyanin homologs, also contained within red cabbage, can be utilized to optimize P2 production3. To this end, Denish et al. found an enzyme, 1AUR, that they modified with the mutation M73H, enabling the enzyme to deacetylate the P3-P8 anthocyanins, converting P3-P8 into the P2 anthocyanin3. Although future studies must be undertaken to prove stability and safety, this anthocyanin converting enzyme might be a possible alternative route in obtaining a natural Blue No. 1.
The search for a natural Blue No. 1 alternative may have reached an end with the discovery of the 1AUR enzyme capable of converting anthocyanins into the appropriately brilliantly blue homolog. Finding the path towards a natural, true-blue dye might be the influence needed to encourage a full switch from artificial to natural food coloring additives in manufacturing practices. Although a natural Blue No. 1 may still be years away, the use of anthocyanins as natural color additives might reach greater influence and open the door for the development of more natural color additives.
References
- https://www.fda.gov/industry/color-additives/color-additives-history
- https://www.acs.org/content/acs/en/education/resources/highschool/chemmatters/past-issues/2015-2016/october-2015/food-colorings.html?inline-read-more
- Denish, Pamela R., et al. “Discovery of a natural cyan blue: A unique food-sourced anthocyanin could replace synthetic brilliant blue.” Science Advances 7.15 (2021): eabe7871.
- Wollen, Malia. “The Quest to Make a True Blue M&M.” The New York Times Magazine, 5 Oct. 2016, www.nytimes.com/interactive/2016/10/09/magazine/blue-food-coloring-mars-company.html.
- https://natcol.org/library/what-are-natural-food-colours/
- https://www.bbvaopenmind.com/en/science/research/mauve-the-history-of-the-colour-that-revolutionized-the-world/
- https://line.17qq.com/articles/offijofz_p3.html