By Brianna Evans
Dr. Joy DeGruy coined the theory Post Traumatic Slave Syndrome (PTSS) and defines it as, “a condition that exists when a population has experienced multigenerational trauma resulting from centuries of slavery and continues to experience oppression and institutionalized racism today.”1 Is it possible that trauma can be passed down generations? Many diseases are known to have a strong genetic component. Some diseases (like Lynch syndrome) have high penetrance and familial history (and cause rapid and profuse development of intestinal polyps by the age of 50), while others, (like metabolic syndrome, a combination of high blood sugar, high blood pressure, abnormal cholesterol levels, and excess body fat around waist), rely more strongly on environmental influences. Even with psychological disorders like addiction and depression, we know that there are genetic and environmental risk factors. Knowing that many diseases can be passed on from parent to offspring, it is possible that trauma can also be passed down generations. When studying generational trauma, scientists look toward epigenetic changes including DNA methylation, histone modification, and elevated or reduced expression of a non-coding RNA (ncRNA) important in regulating gene transcription.2
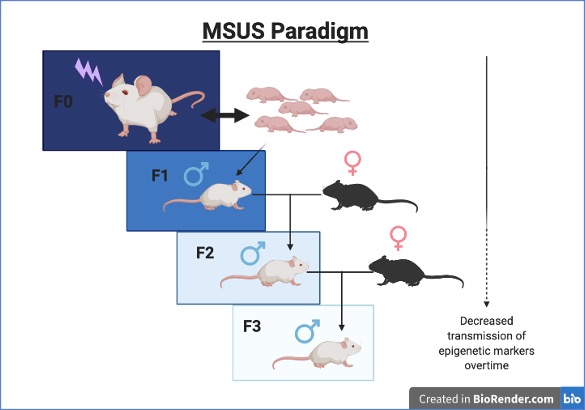
Epigenetic modifications do not change the genetic sequence, but can alter gene expression. One common way to study epigenetic changes in the context of trauma is through mice exposed to chronic, unpredictable maternal separation and maternal stress (MSUS) as shown in Figure 1. MSUS mice have progeny that demonstrate depressive-like behaviors responsive to the antidepressant desipramine.3 Along with increased depressive-like symptoms, MSUS mice progeny have hypermethylation of MeCP2 (methyl CpG binding protein 2) and CB1 (cannabinoid-1) receptor and hypomethylation of CRFR2 (Corticotropin-releasing factor receptor 2).3 Hypermethylation of MeCP2 and CB1 are associated with increased stress response and emotionality respectively, while hypomethylation of CRFR2 leads to reduced expression of CRFR2 receptor.3
Modification of histones open or close off certain areas on chromosomes, altering the ability of genes in those regions to be transcribed and expressed. MSUS mice also express histone modifications on the mineralocorticoid receptor gene (MR) associated with reduced MR expression.4 This finding is important since decreased MR is implicated in depressive disorders, and correlates with a higher incidence of suicide.5 Regardless of the means of MR modulation, either through exposure to receptor agonist, epigenetic modifications or within the MSUS model, its inhibition results in decreased fear and avoidance response.4 MR expression changes brought on by epigenetic alterations resulting in the same effect as MR targeted therapy further demonstrates MR expression association with behavior. ncRNAs are RNAs that are not translated into protein and regulate gene expression during and post-translation. One type of ncRNA, microRNA (miRNA), frequently plays a role in diseases, yet are often still poorly understood.2 In addition to epigenetic modifications, there has been research on several changes in the expression of miRNA. Important for the support of inherited stress, injection of miRNA isolated from MSUS mice sperm into young mice resulted in development of behavioral and metabolic effects not seen in control littermates.6
There are also human studies that demonstrate methylation changes and their correlation with behavioral developments. Offspring of pregnant women during the Tutsi genocide had higher levels of PTSD and depression, and demonstrated higher methylation levels of NR3C1 (Nuclear receptor subfamily 3 group c member 1), a glucocorticoid receptor.7 The increased methylation of NR3C1 was associated with lower cortisol levels and PTSD.8 Lower cortisol levels are associated with PTSD most likely due to increased glucocorticoid receptor sensitivity to steroid ligands.9 Additionally, parents who experienced the holocaust had higher methylation of FKBP5, a moderator of glucocorticoid activity, while their offspring had lower levels of FKBP5 methylation.10 Less FKBP5 methylation can be attributed to accommodation of the offspring to handle similar starved conditions as their parents did during the holocaust, which would help them store fat easier. Unfortunately, less methylation of FKBP5 increased the offspring’s risk for metabolic syndrome and obesity.11 Since the offspring are growing up in a period where food is more easily accessible and life is more sedentary, it is much easier for them to gain weight and very difficult for them to lose it.
There are, however, limitations with researching generational trauma in humans. The research is observational and retrospective, using pre-existing trauma groups to examine differences in gene expression and behavior. Retrospective observational studies have risks of recall bias (subjects may not recall an event as clearly), confounding, and selection bias. The randomized control trial is the golden standard of research, but purposefully traumatizing people in order to study their epigenetic changes and offspring is unethical. Also, one could argue that it is the environment, growing up with a stressed parent, that can cause these epigenetic changes. Moreover, human generational trauma studies have low sample size and a lack of longitudinal data collection beyond two generations. As a result, it is not known if these changes remain heritable after two generations. Nevertheless, human generational trauma studies give some evidence that inheriting trauma may occur in humans.
If trauma can be generationally inherited, what does this mean for descendants of American slavery? Mouse studies show that a generation exposed to a trauma has genetic and behavioral changes that pass down two generations, albeit in a gradient fashion decreasing as the generations progressed (Figure 1).2,3,6 Notably, the traumatized mice were bred with wild-type mice every generation, so the trauma did not continue to build up for several generations, allowing alleviation of stress, and introduction of non-traumatic epigenetic patterns.2,3,6 When it comes to American slavery, however, multiple generations went through this trauma, and changes may compound and worsen. This introduces many more questions we yet do not understand: Could this make epigenetic changes from trauma to be more permanent? How have other traumatic periods for African Americans like the Jim Crow era contributed to generational trauma as well? How do impacts of current injustices (such as police brutality) influence epigenetics in people today? The animal and human studies on generational trauma do not give us solid proof for Dr. DeGruy’s PTSS theory, however, they do show that PTSS could be a true problem within the African American community. Studying PTSS and other forms of generational trauma will hopefully aid in finding treatments and therapies for those still struggling with ancestral trauma.
Trauma impacts individuals in multiple ways. You can learn more about symptoms of trauma here (https://www.helpguide.org/articles/ptsd-trauma/coping-with-emotional-and-psychological-trauma.htm). If you are experiencing distress, please know that there is help available. Please use the following resources:
- Crisis Textline Text HOME to 741741
- National Suicide Prevention Lifeline Call 800-273-8255 or Chat with Lifeline
- Veterans Crisis Line (VA) Call 800-273-8255 or text 838255
- Physician Support Line Call 1-888-409-0141
- NAMI Helpline: 800-950-6264 M-F, 10 a.m. – 6 p.m., ET
References
- DeGruy, Joy. (2018). Post traumatic slave syndrome: America’s legacy of enduring injury and healing. United States: Joy DeGruy Publications.
- Klengel T, Dias BG, Ressler KJ. Models of Intergenerational and Transgenerational Transmission of Risk for Psychopathology in Mice. Neuropsychopharmacology. 2016 Jan;41(1):219-31. doi: 10.1038/npp.2015.249. Epub 2015 Aug 18. PMID: 26283147; PMCID: PMC4677139.
- Franklin TB, Russig H, Weiss IC, Gräff J, Linder N, Michalon A, Vizi S, Mansuy IM. Epigenetic transmission of the impact of early stress across generations. Biol Psychiatry. 2010 Sep 1;68(5):408-15. doi: 10.1016/j.biopsych.2010.05.036. Epub 2010 Jul 31. PMID: 20673872.
- Gapp, K., Soldado-magraner, S., Alvarez-sánchez, M., Bohacek, J., Vernaz, G., Shu, H., Mansuy, I. M. (2014). Early life stress in fathers improves behavioural flexibility in their offspring. Nature Communications, 5, 5466. doi:http://dx.doi.org.ezaccess.libraries.psu.edu/10.1038/ncomms6466
- López JF, Chalmers DT, Little KY, Watson SJ. A.E. Bennett Research Award. Regulation of serotonin1A, glucocorticoid, and mineralocorticoid receptor in rat and human hippocampus: implications for the neurobiology of depression. Biol Psychiatry. 1998 Apr 15;43(8):547-73. doi: 10.1016/s0006-3223(97)00484-8. PMID: 9564441.
- Gapp, K., Jawaid, A., Sarkies, P., Bohacek, J., Pelczar, P., Prados, J., Farinelli, L., Miska, E., Mansuy, I.M., 2014. Implication of sperm RNAs in transgenerational inheritance of the effects of early trauma in mice. Nature Neuroscience 17, 667–669.. doi:10.1038/nn.3695
- Perroud N., Rutembesa E., Paoloni-Giacobino A., Mutabaruka J., Mutesa L., Stenz L., Malafosse A., Karege F. The Tutsi genocide and transgenerational transmission of maternal stress: Epigenetics and biology of the HPA axis. World J. Biol. Psychiatry. 2014;15:334–345. doi: 10.3109/15622975.2013.866693.
- Youssef, N. A., Lockwood, L., Su, S., Hao, G., & Rutten, B. (2018). The Effects of Trauma, with or without PTSD, on the Transgenerational DNA Methylation Alterations in Human Offsprings. Brain sciences, 8(5), 83. https://doi.org/10.3390/brainsci8050083
- Rachel Yehuda, Jonathan Seckl, Minireview: Stress-Related Psychiatric Disorders with Low Cortisol Levels: A Metabolic Hypothesis, Endocrinology, Volume 152, Issue 12, 1 December 2011, Pages 4496–4503, https://doi.org/10.1210/en.2011-1218
- Yehuda R., Bierer L.M. Transgenerational transmission of cortisol and PTSD risk. Prog. Brain Res. 2008;167:121–135.
- Yehuda R., Daskalakis N.P., Bierer L.M., Bader H.N., Klengel T., Holsboer F., Binder E.B. Holocaust Exposure Induced Intergenerational Effects on FKBP5 Methylation. Biol. Psychiatry. 2016;80:372–380. doi: 10.1016/j.biopsych.2015.08.005.
Pingback: Generational Trauma and Family Curses - Malum In Se
Pingback: Generational Trauma & PTSD Within the Black Community – Break free of Addiction
Pingback: Generational Trauma & PTSD Within the Black Community
Pingback: The Importance Of Black History In Understanding The Police Murder Of Tamir Rice - Black Star News
Pingback: Generational Trauma & PTSD Within the Black Community - Meadows Behavioral Healthcare
이건 정말 훌륭한 자료인데, 무료로 제공하시다니. 양질의 자료를 무료로 제공하는 것의 가치를 이해하는 블로그를 보는 게 좋아요.