By Nicole Lookfong
The COVID-19 pandemic has created psychological consequences within the population, such as an increased risk for developing post-traumatic stress disorder (PTSD), by way of chronic stress, increased perception of threats, rising mortality rate, and limited resources1. Normally, the prevalence of PTSD in the United States is 7-8%, with individuals experiencing symptoms such as avoidance, intrusive thoughts, negative alterations in cognition or mood, arousal, and reactivity symptoms2,3. In the midst of the COVID-19 outbreak in June of 2020, the CDC released a survey that showed trauma/stressor-related disorder symptoms had risen by 26% because of the pandemic1. In particular, emergency healthcare workers are becoming more susceptible to mental health problems, as prior to COVID-19, first-line responders’ risk of developing PTSD ranged from 10%-20%, and the risk was higher within the intensive care unit at 8%-30%4,5,6,7,8,9,10. These healthcare workers’ risk for PTSD can increase during pandemics due to increased patient flow, high daily fatality rates, physical isolation to protect patients, and a high risk of contamination11,12,13. Understanding the biological mechanism of PTSD and how it translates to behavioral symptoms is crucial to inhibit the severity of its progression.
One way to study the mechanism of PTSD is using fear conditioning in animal models, a form of classical conditioning that takes advantage of highly adaptive natural learning processes to generate fear memories towards normally unthreatening stimuli14. In fear conditioning, an unconditioned stimulus is paired with a neutral stimulus, allowing individuals to avoid aversive events by remembering the event’s contextual cues14. For example, picture a mouse hearing an auditory tone and receiving a foot shock at the same time (fig 1) 14. The auditory tone in this case is the neutral stimulus or the contextual cue, while the foot shock is the unconditioned stimulus or the traumatic event14. The mouse will start to associate the unconditioned stimulus (the foot shock) with the neutral stimulus (the auditory tone), forming a fear memory that results in the unconditioned stimulus (the foot shock) becoming a conditioned stimulus (fig 1)14. A conditioned stimulus is a specific context or cue that produces a response with the same physiological and behavioral effects as the unconditioned stimulus (foot shock), long after the trauma itself14. Individuals with PTSD may be aware of their conditioned stimuli, such as combat veterans who are reminded of potential bombs by everyday objects like unmarked packages14. However, those same veterans may see sudden movement in their periphery and experience a fear response they didn’t know they had, becoming hypervigilant to triggers and reminders of their trauma in order to avoid them14. Due to COVID-19, we wonder what contextual cues may become triggers for individuals who develop PTSD from the pandemic.
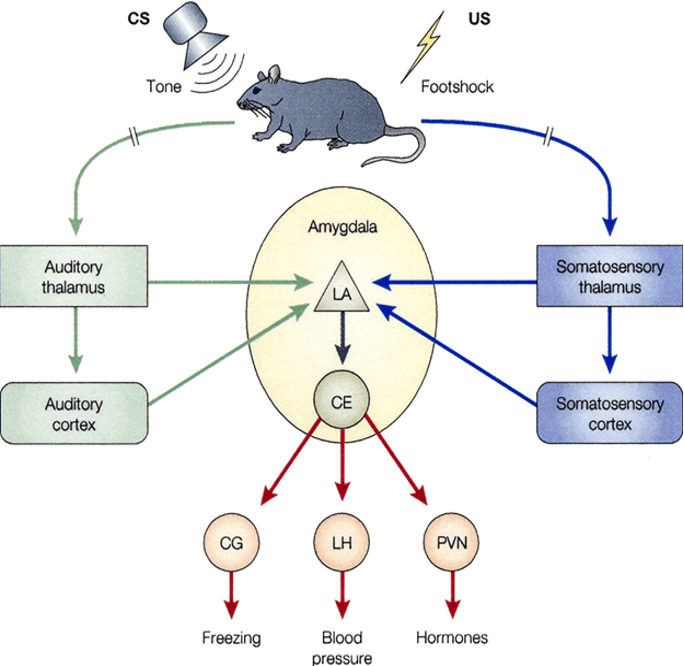
A neutral stimulus (auditory tone) temporally associated with an aversive event (foot shock) forms a conditioned stimulus. Information about the tone and the foot shock travel through the appropriate sensory pathways converging in the amygdala. The fear signals then travel from the amygdala to many different parts of the brain, resulting in the expression of the fear responses such as freezing behavior, increased blood pressure and heart rate, and secretion of pituitary-adrenal hormones. Abbreviations: CS (conditioned stimulus), US (unconditioned stimulus), LA (lateral amygdala), CE (central amygdala), CG (central gray), LH (lateral hypothalamus), PVN (paraventricular hypothalamus). Modified from Phelps & LeDoux, 2005 (15).
To understand how contextual cues become triggers, it is important to look at the neurophysiology behind PTSD. The amygdala, which is the primary region for fear and stress, has been extensively studied to understand mechanisms of fear memory formation in PTSD research16,17. In the above example of the mouse who received a foot shock and auditory tone, sensory information about the neutral stimulus (auditory tone) and unconditioned stimulus (foot shock) enters the amygdala from sensory processing areas (fig. 1)16. While in the amygdala, the information about the neutral stimulus (auditory tone) and unconditioned stimulus (foot shock) converge, producing a memory of the conditioned stimulus (tone anticipating foot shock) (fig. 1)17. The amygdala then sends fear signals to the areas of the brain that regulate the fear memory’s behavioral and physiological consequences (fig. 1)17. Prolonged stress from the hyperactive behavioral and physiological response can lead to maladaptive changes to the hypothalamo-pituitary-adrenocortical (HPA) axis, leading to the development of PTSD18.
The HPA axis regulates the body’s response to stress through an increased release of glucocorticoids such as cortisol (fig. 2)18. Outputs from the amygdala stimulate corticotropin-releasing factor neurons in the hypothalamus, which results in the release of cortisol from the adrenal glands (fig. 2)18,20. When a certain threshold is reached, cortisol can provide negative feedback to the HPA axis telling it to shut down the production of cortisol (fig. 2)21,22. Individuals with PTSD can have a dysregulated HPA axis with varying cortisol levels.23 Animal studies and postmortem studies show that early childhood trauma causes methylation on the glucocorticoid receptors in the hippocampus, which decreases the inhibition of the HPA axis stress response24. Disruption of HPA axis function in PTSD results in an inability to inhibit other stress response circuits, leading to elevated levels of the neurotransmitter and hormone norepinephrine, triggering a fight-or-flight response25. Norepinephrine can then project to various brain areas, including the amygdala, HPA axis, and prefrontal cortex, to interact with corticotropin-releasing factor neurons26. As a result, fear memories are acquired and consolidated, leading to increased stress response reactivity and PTSD symptoms including hypervigilance and arousal26.

HPA axis’s stress response begins when corticotropin releasing hormone neurons are secreted from the hypothalamus. The corticotropin releasing hormones then activate the anterior pituitary, stimulating the release of adrenocorticotropic hormone. As adrenocorticotropic hormone circulates through the system, synthesizing cortisol. Cortisol then travels through the bloodstream affecting different organs and preparing the system for “fight or flight”. Abbreviations: CRH (corticotropin-releasing hormone), ACTH (adrenocorticotropic hormone), CORT (cortisol). Modified from Herman et al., 2016 (19).
In addition to regulation of the HPA axis by cortisol and norepinephrine, the HPA axis is also regulated by the prefrontal cortex (PFC) and the hippocampus18,27. Both regions are crucial for modulating the stress response and regulating PTSD symptoms18,27. In the PFC, the dorsolateral and ventromedial regions work together to format abstract thought and goal-directed behavior27. However, when activity in the PFC is altered by chronic or traumatic stress, individuals can experience intrusive thoughts and fear responses, resulting in problems with attention and appropriate emotion regulation28,29,30. PTSD severity has been shown to increase when ventromedial PFC activity decreases, as the ventromedial PFC activity is inversely related to amygdala activity, leading to dysregulation of the HPA axis (fig. 3)31. Similar to the PFC, the hippocampus also allows for modulation of the HPA axis through amygdala inhibition18. Individuals with PTSD have decreased hippocampal volumes, and while it is unclear whether this reduced size occurs before or after the trauma, individuals with smaller hippocampal volumes are at increased risk of developing PTSD32,33,34. Since the hippocampus is significant for pattern separation, memory formation, and cognition35,36, individuals with PTSD have difficulty distinguishing between recalled traumatic memories and similar safe environments, resulting in overgeneralization of traumatic events to benign ones37.
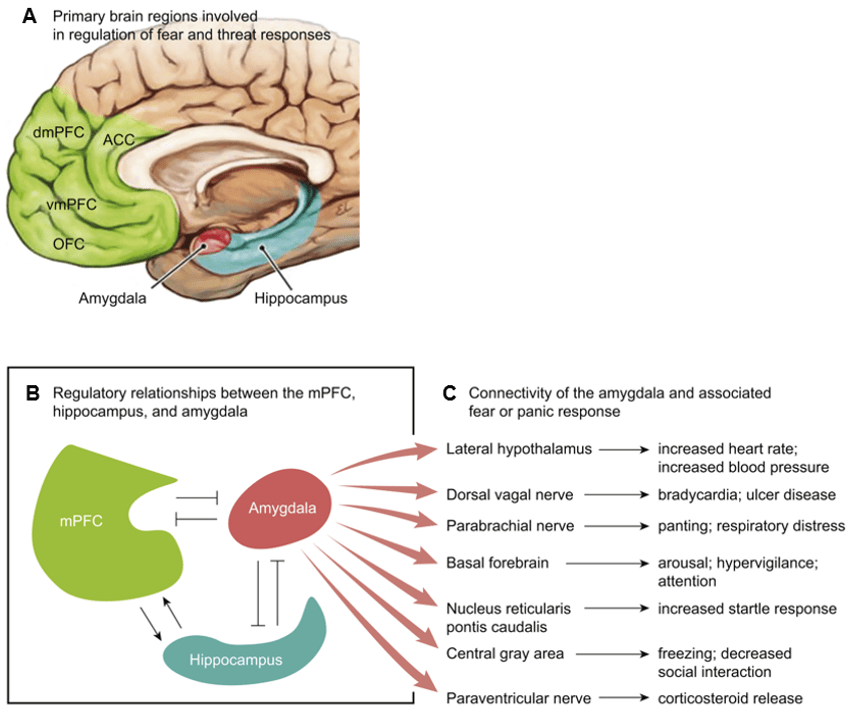
A) Fundamental brain regions for regulating fear and threat responses are the amygdala, hippocampus, and ventromedial prefrontal cortex. B) and C) The medial prefrontal cortex and hippocampus regulate amygdala activity, fear modulation, and context. The ventromedial prefrontal cortex reduces subjective distress when inhibiting amygdala activity. The hippocampus encodes fear memories. Abbreviations: dmPFC (dorsomedial prefrontal cortex), ACC (anterior cingulate cortex), vmPFC (ventromedial prefrontal cortex), OFC (orbitofrontal cortex), mPFC (medial prefrontal cortex). Modified from Ross et al., 2017 (14).
While traumatic memories cannot be removed, they can be altered, and new inhibitory memories can be made to outcompete them14,38. Many current PTSD therapeutic treatments are focused on extinction, which is the recall of a long-term memory into working memory, allowing the memory to be updated with new information before it can be reconsolidated14,39. Extinction is achieved through repeated exposure to a conditioned stimulus in the absence of the unconditioned stimulus until the conditioned fear response is effectively reduced14. For a mouse conditioned to associate an auditory tone with a foot shock, extinction would involve repeatedly exposing the mouse to the auditory tone without the foot shock until the fear response subsides. If an individual with PTSD recalls a traumatic memory and re-experiences it, the memory or their interpretation of the memory can be updated to include added feelings of guilt, shame, and emotional valence38. However, when a recalled memory is updated with new information, exposure therapy can allow for extinction where the memory elicits no adverse consequences, resulting in an adaptive response14,40,41. However, studies have found that extinction is not always effective, as the fear memory can return following a certain amount of time, a change in contexts, or the reintroduction of the unconditioned stimulus14,40,41. Therefore, it is important to use extinction in multiple contexts and replace the unconditioned stimulus with non-aversive stimuli42,43. In addition to behavioral therapy, numerous pharmacologic approaches are being developed to help improve fear-memory extinction training44,45.
There is still more to learn about the underlying pathophysiological processes of PTSD that create aberrant behavior and dysfunction in the neurocircuits and their connections. Neuroscientists will lead us to a better understanding of these underlying processes and allow us to develop and improve treatments. With COVID-19 causing excessive stress in people’s lives, it is imperative that we better understand PTSD to help those who struggle to adjust to normative life once the pandemic passes.
Nicole Lookfong is an undergraduate at Penn State Harrisburg majoring in genetics and developmental biology and is a lab technician in Dr. Yuval Silberman’s lab at Penn State College of Medicine. Nicole was mentored by Mariam Melkumyan, one of our Lions Talk Science Editors, to write this article.
References
[1] M. É. Czeisler, R. I. Lane, E. Petrosky, J. F. Wiley, A. Christensen, R. Njai, M. D.
Weaver, R. Robbins, E. R. Facer-Childs, L. K. Barger, C. A. Czeisler, M. E. Howard, and S. M. Rajaratnam, “Mental Health, Substance Use, and Suicidal Ideation During the COVID-19 Pandemic — United States, June 24–30, 2020,” MMWR. Morbidity and Mortality Weekly Report, vol. 69, no. 32, pp. 1049–1057, Aug. 2020.
[2] Desk reference to the diagnostic criteria from DSM-5. Arlington, VA: American
Psychiatric Association, 2016.
[3] “How Common is PTSD in Adults?,” U.S. Department of Veterans Affairs, 13-Sep-2018.
[Online]. Available: https://www.ptsd.va.gov/understand/common/common_adults.asp. [Accessed: 15-Jan-2021].
[4] S. Clohessy and A. Ehlers, “PTSD symptoms, response to intrusive memories and coping
in ambulance service workers,” British Journal of Clinical Psychology, vol. 38, no. 3, pp. 251–265, Dec. 1999.
[5] J. DeLucia, C. Bitter, J. Fitzgerald, M. Greenberg, P. Dalwari, and P. Buchanan,
“Prevalence of Post-Traumatic Stress Disorder in Emergency Physicians in the United States,” Western Journal of Emergency Medicine, Volume 20, Issue 5 Western Journal of Emergency Medicine, vol. 20, no. 5, pp. 740–746, Aug. 2019.
[6] F. Grevin, “Posttraumatic Stress Disorder, Ego Defense Mechanisms, and Empathy
among Urban Paramedics,” Psychological Reports, vol. 79, no. 2, pp. 483–495, Oct. 1996.
[7] M. Karanikola, M. Giannakopoulou, M. Mpouzika, C. P. Kaite, G. Z. Tsiaousis, and E. D. E. Papathanassoglou, “Dysfunctional psychological responses among Intensive Care Unit nurses: a systematic review of the literature,” Revista da Escola de Enfermagem da USP, vol. 49, no. 5, pp. 847–857, 2015.
[8] D. A. Machado, N. M. A. D. Figueiredo, L. D. S. Velasques, C. A. D. M. Bento, W. C. A. Machado, and L. A. M. Vianna, “Cognitive changes in nurses working in intensive care units,” Revista Brasileira de Enfermagem, vol. 71, no. 1, pp. 73–79, 2018.
[9] M. Mealer, E. L. Burnham, C. J. Goode, B. Rothbaum, and M. Moss, “The prevalence
and impact of post traumatic stress disorder and burnout syndrome in nurses,” Depression and Anxiety, vol. 26, no. 12, pp. 1118–1126, Nov. 2009.
[10] N. Robertson and A. Perry, “Institutionally based health care workers’ exposure to
traumatogenic events: Systematic review of PTSD presentation,” Journal of Traumatic Stress, May 2010.
[11] W. Berger, E. S. F. Coutinho, I. Figueira, C. Marques-Portella, M. P. Luz, T. C. Neylan,
C. R. Marmar, and M. V. Mendlowicz, “Rescuers at risk: a systematic review and meta-regression analysis of the worldwide current prevalence and correlates of PTSD in rescue workers,” Social Psychiatry and Psychiatric Epidemiology, vol. 47, no. 6, pp. 1001–1011, Jun. 2011.
[12] S. K. Brooks, G. J. Rubin, and N. Greenberg, “Traumatic stress within disaster-exposed
occupations: overview of the literature and suggestions for the management of traumatic stress in the workplace,” British Medical Bulletin, vol. 129, no. 1, pp. 25–34, Nov. 2018.
[13] K. Petrie, J. Milligan-Saville, A. Gayed, M. Deady, A. Phelps, L. Dell, D. Forbes, R. A.
Bryant, R. A. Calvo, N. Glozier, and S. B. Harvey, “Prevalence of PTSD and common mental disorders amongst ambulance personnel: a systematic review and meta-analysis,” Social Psychiatry and Psychiatric Epidemiology, vol. 53, no. 9, pp. 897–909, Jun. 2018.
[14] D. A. Ross, M. R. Arbuckle, and M. J. Travis, “An Integrated Neuroscience Perspective
on Formulation and Treatment Planning for Posttraumatic Stress Disorder,” JAMA Psychiatry, vol. 74, no. 4, pp. 407–415, Apr. 2017.
[15] E. A. Phelps and J. E. Ledoux, “Contributions of the Amygdala to Emotion Processing: From Animal Models to Human Behavior,” Neuron, vol. 48, no. 2, pp. 175–187, Oct. 2005.
[16] J. Ledoux, C. Farb, and D. Ruggiero, “Topographic organization of neurons in the
acoustic thalamus that project to the amygdala,” The Journal of Neuroscience, vol. 10, no. 4, pp. 1043–1054, 1990.
[17] J. F. Medina, J. C. Repa, M. D. Mauk, and J. E. Ledoux, “Parallels between cerebellum-
and amygdala-dependent conditioning,” Nature Reviews Neuroscience, vol. 3, no. 2, pp. 122–131, Feb. 2002.
[18] B. Myers, J. M. Mcklveen, and J. P. Herman, “Neural Regulation of the Stress Response:
The Many Faces of Feedback,” Cellular and Molecular Neurobiology, vol. 32, no. 5, pp. 683–694, Feb. 2012.
[19] J. P. Herman, J. M. Mcklveen, S. Ghosal, B. Kopp, A. Wulsin, R. Makinson, J. Scheimann, and B. Myers, “Regulation of the Hypothalamic-Pituitary-Adrenocortical Stress Response,” Comprehensive Physiology, pp. 603–621, Mar. 2016.
[20] J. Spiess, J. Rivier, C. Rivier, and W. Vale, “Primary structure of corticotropin-releasing
factor from ovine hypothalamus.,” Proceedings of the National Academy of Sciences, vol. 78, no. 10, pp. 6517–6521, Oct. 1981.
[21] F. L. Groeneweg, H. Karst, E. R. D. Kloet, and M. Joëls, “Mineralocorticoid and
glucocorticoid receptors at the neuronal membrane, regulators of nongenomic corticosteroid signalling,” Molecular and Cellular Endocrinology, vol. 350, no. 2, pp. 299–309, Jun. 2011.
[22] E. R. D. Kloet, M. Joëls, and F. Holsboer, “Stress and the brain: from adaptation to
disease,” Nature Reviews Neuroscience, vol. 6, no. 6, pp. 463–475, May 2005.
[23] R. Yehuda, “Status of Glucocorticoid Alterations in Post-traumatic Stress Disorder,”
Annals of the New York Academy of Sciences, vol. 1179, no. 1, pp. 56–69, Oct. 2009.
[24] P. O. Mcgowan, A. Sasaki, A. C. D’alessio, S. Dymov, B. Labonté, M. Szyf, G. Turecki,
and M. J. Meaney, “Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse,” Nature Neuroscience, vol. 12, no. 3, pp. 342–348, Mar. 2009.
[25] L. W. Hawk, A. L. Dougall, R. J. Ursano, and A. Baum, “Urinary Catecholamines and
Cortisol in Recent-Onset Posttraumatic Stress Disorder After Motor Vehicle Accidents,” Psychosomatic Medicine, vol. 62, no. 3, pp. 423–434, 2000.
[26] K. Snyder, W.-W. Wang, R. Han, K. Mcfadden, and R. J. Valentino,
“Corticotropin-Releasing Factor in the Norepinephrine Nucleus, Locus Coeruleus, Facilitates Behavioral Flexibility,” Neuropsychopharmacology, vol. 37, no. 2, pp. 520–530, Oct. 2011.
[27] A. F. Arnsten, M. A. Raskind, F. B. Taylor, and D. F. Connor, “The effects of stress
exposure on prefrontal cortex: Translating basic research into successful treatments for post-traumatic stress disorder,” Neurobiology of Stress, vol. 1, pp. 89–99, Jan. 2015.
[28] J. Barrash, D. Tranel, and S. W. Anderson, “Acquired Personality Disturbances
Associated With Bilateral Damage to the Ventromedial Prefrontal Region,” Developmental Neuropsychology, vol. 18, no. 3, pp. 355–381, 2000.
[29] S. L. Thompson-Schill, J. Jonides, C. Marshuetz, E. E. Smith, M. D’esposito, I. P. Kan, R. T. Knight, and D. Swick, “Effects of frontal lobe damage on interference effects in working memory,” Cognitive, Affective, & Behavioral Neuroscience, vol. 2, no. 2, pp. 109–120, Jun. 2002.
[30] A. Wilkins, T. Shallice, and R. Mccarthy, “Frontal lesions and sustained attention,”
Neuropsychologia, vol. 25, no. 2, pp. 359–365, 1987.
[31] L. M. Shin, S. P. Orr, M. A. Carson, S. L. Rauch, M. L. Macklin, N. B. Lasko, P. M.
Peters, L. J. Metzger, D. D. Dougherty, P. A. Cannistraro, N. M. Alpert, A. J. Fischman, and R. K. Pitman, “Regional Cerebral Blood Flow in the Amygdala and Medial PrefrontalCortex During Traumatic Imagery in Male and Female Vietnam Veterans With PTSD,” Archives of General Psychiatry, vol. 61, no. 2, p. 168, Feb. 2004.
[32] B. Roozendaal and J. L. Mcgaugh, “Memory modulation.,” Behavioral Neuroscience,
vol. 125, no. 6, pp. 797–824, 2011.
[33] J. D. Bremner, M. Vythilingam, E. Vermetten, S. M. Southwick, T. Mcglashan, A.
Nazeer, S. Khan, L. V. Vaccarino, R. Soufer, P. K. Garg, C. K. Ng, L. H. Staib, J. S. Duncan, and D. S. Charney, “MRI and PET Study of Deficits in Hippocampal Structure and Function in Women With Childhood Sexual Abuse and Posttraumatic Stress Disorder,” American Journal of Psychiatry, vol. 160, no. 5, pp. 924–932, May 2003.
[34] M. W. Gilbertson, M. E. Shenton, A. Ciszewski, K. Kasai, N. B. Lasko, S. P. Orr, and R.
K. Pitman, “Smaller hippocampal volume predicts pathologic vulnerability to psychological trauma,” Nature Neuroscience, vol. 5, no. 11, pp. 1242–1247, Oct. 2002.
[35] N. S. Burghardt, E. H. Park, R. Hen, and A. A. Fenton, “Adult-born hippocampal neurons
promote cognitive flexibility in mice,” Hippocampus, vol. 22, no. 9, pp. 1795–1808, Sep. 2012.
[36] C. D. Clelland, M. Choi, C. Romberg, G. D. Clemenson, A. Fragniere, P. Tyers, S.
Jessberger, L. M. Saksida, R. A. Barker, F. H. Gage, and T. J. Bussey, “A Functional Role for Adult Hippocampal Neurogenesis in Spatial Pattern Separation,” Science, vol. 325, no. 5937, pp. 210–213, Jul. 2009.
[37] J. F. Medina, J. C. Repa, M. D. Mauk, and J. E. Ledoux, “Parallels between cerebellum-
and amygdala-dependent conditioning,” Nature Reviews Neuroscience, vol. 3, no. 2, pp. 122–131, Feb. 2002.
[38] D. A. Ross, M. J. Travis, and M. R. Arbuckle, “Posttraumatic Stress Disorder in a Young
Adult Military Veteran,” JAMA Psychiatry, vol. 74, no. 4, pp. 417–418, Apr. 2017.
[39] K. M. Myers and M. Davis, “Behavioral and Neural Analysis of Extinction,” Neuron,
vol. 36, no. 4, pp. 567–584, Nov. 2002.
[40] M. E. Bouton, “Context, ambiguity, and unlearning: sources of relapse after behavioral
extinction,” Biological Psychiatry, vol. 52, no. 10, pp. 976–986, Nov. 2002.
[41] M. E. Bouton, “Context and ambiguity in the extinction of emotional learning:
Implications for exposure therapy,” Behaviour Research and Therapy, vol. 26, no. 2, pp. 137–149, 1988.
[42] J. E. Dunsmoor, F. Ahs, D. J. Zielinski, and K. S. Labar, “Extinction in multiple virtual
reality contexts diminishes fear reinstatement in humans,” Neurobiology of Learning and Memory, vol. 113, pp. 157–164, 2014.
[43] J. E. Dunsmoor, V. D. Campese, A. O. Ceceli, J. E. Ledoux, and E. A. Phelps,
“Novelty-Facilitated Extinction: Providing a Novel Outcome in Place of an Expected Threat Diminishes Recovery of Defensive Responses,” Biological Psychiatry, vol. 78, no. 3, pp. 203–209, Aug. 2015.
[44] M. Kindt, M. Soeter, and B. Vervliet, “Beyond extinction: erasing human fear responses
and preventing the return of fear,” Nature Neuroscience, vol. 12, no. 3, pp. 256–258, Mar. 2009.
[45] L. Ledgerwood, R. Richardson, and J. Cranney, “d-cycloserine facilitates extinction of
learned fear: Effects on reacquisition and generalized extinction,” Biological Psychiatry, vol. 57, no. 8, pp. 841–847, Apr. 2005.